Clinical Medicine Experimental Medicine Management
|
|
|
- Kajetan Kowal
- 10 lat temu
- Przeglądów:
Transkrypt
1 / MARCH 2015 ISSN MedicalPROBLEMS PUBLIC HEALTH Secretary of State, dr n. med Beata Małecka-Libera, on creating a healthier nation Clinical Experimental Management Peer-reviewed articles about the latest developments in medicine, healthcare, ethics and clinical leadership, released by the Central Clinical Hospital of the Ministry of the Interior in Warsaw. Academic biomedical research papers to support clinical practice and help develop diagnostic and therapeutic methods and techniques, released by the Polish Academy of Sciences. Public and private sector managers address today s challenges and offer insights into best practice and how to achieve effective governance and operational improvements in the healthcare sector. CENTRAL CLINICAL HOSPITAL OF THE MINISTRY OF THE INTERIOR, WARSAW
2 PROGRAM KONFERENCJI III KONFERENCJA NAUKOWO-SZKOLENIOWA PRZESZCZEPIANIE NARZĄDÓW UNACZYNIONYCH r. 8:15-9:00 Rejestracja uczestników 9:00-9:20 Inauguracja Konferencji BLOK A SESJA I PRZESZCZEPIANIE NARZĄDÓW UNACZYNIONYCH 9:20-9:40 I Wykład Prof. Roman Danielewicz Stan polskiej transplantologii na tle Europy i świata 9:40-10:00 II Wykład Prof. Marek Durlik Przeszczepienie trzustki doświadczenia własne ośrodka 10:00-10:20 III Wykład Prof. Zbigniew Włodarczyk Przeszczepianie nerki doświadczenia własne ośrodka 10:20-10:40 IV Wykład Prof. Krzysztof Zieniewicz Przeszczepienie wątroby doświadczenia własne ośrodka wątroby 10:40-11:00 V Wykład Prof. Jarosław Czerwiński Dawca trzustki kryteria kwalifikacji, czy powinniśmy je zawęzić? 11:00-11:20 PRZERWA KAWOWA BLOK B SESJA II POWIKŁANIA W TRANSPLANTOLOGII 11:20-11:40 VI Wykład Prof. Lech Cierpka Powikłania naczyniowe u biorców przeszczepów unaczynionych wątroby, trzustki, nerki 11:40-12:00 VII Wykład Prof. Magdalena Durlik Powikłania wirusowe po transplantacji 12:00-12:20 VIII Wykład Prof. Bolesław Rutkowski Powikłania bakteryjne po transplantacji 12:20-12:35 IX Wykład Inwazyjne zakażenia grzybicze u pacjenta po transplantacji firma MSD 12:35-12:50 X Wykład Profilaktyka zakażeń grzybiczych w świetle refundacji Ministerstwa Zdrowia firma MSD 12:50-13:20 PRZERWA KAWOWA BLOK C SESJA III ŻYWY DAWCA NERKI 13:20-13:40 XI wykład Prof. Zbigniew Gaciong Rozwój i efekty programu Żywy Dawca Nerki w Polsce 13:40-14:00 XII Wykład Prof. Jarosław Czerwiński Dawca nerki do przeszczepu rodzinnego 14:00-14:20 XIII Wykład Indukcja w transplantologii firma Sanofi 14:20-15:20 PRZERWA NA KAWOWA + LUNCH 15:20-15:40 Losowanie nagórd 15:40-16:00 Zakończenie Konferencji wraz z podsumowaniem Zapisy: tel.: (22) , dyrekcja@cskmswia.pl
3 Patronat: III Konferencja Naukowo-Szkoleniowa Fundacji przy Centralnym Szpitalu Klinicznym MSW NOWOTWORY GÓRNEGO ODCINKA UKŁADU POKARMOWEGO Witamy wszystkich uczestników konferencji LECZENIE WIELOSPECJALISTYCZNE NOWOTWORÓW GÓRNEGO ODCINKA UKŁADU POKARMOWEGO 16 kwietnia 2015 r. Aula CSK MSW w Warszawie, ul. Wołoska 137 Kierownictwo naukowe i prowadzenie: Prof. dr hab. med. Marek DURLIK Prof. dr hab. med. Andrzej W. SZAWŁOWSKI From the Editor dr hab. n. med. Marek Durlik prof. PAN Director Central Clinical Hospital of the Ministry of the Interior, Warsaw Welcome to the first issue of Medical Problems for Last year we celebrated our 60th Anniversary of documenting progress and breakthroughs in the medical profession. This year we plan to continue in this tradition. Changes to our healthcare system have been at the forefront of our public consciousness for a while now. Some reforms have been controversial, however this is a common feature of change and we must always maintain our focus on the ultimate goal of helping people lead longer, healthier and happier lives. To this end, the Polish government is currently introducing the first Public Health bill in Parliament. It is a vital piece of legislation. In the United States, public health and preventative healthcare initiatives have added 25 years to the average person s life expectancy. The greatest public health initiatives of the 20th Century include changing public attitudes toward smoking. Let s not forget that little more than 60 years ago, doctors were still promoting cigarettes. Fortunately we live in different times. Immunizations and the control of infectious diseases are further public health achievements. The first breakthroughs in viral isolation and tissue culture occurred on the eve of the last century. They enabled, for example, the treatment of foot and mouth disease. Since then, the dawn of molecular techniques over the last 40 years has paved the way to the treatment of diseases such as Hepatitis C and HIV. Today we face further health challenges, many of them preventable. I am honoured that the Secretary of State, Beata Małecka-Libera, who is responsible for preparing the new Public Health bill, granted us an interview on the subject. We all have a role to play in its implementation. A hospital is a critical public institution. Not only does it treat the sick and contribute to the prevention of disease, but it also plays a key role in research and facilitating dialogue in the medical profession. In April, our hospital s foundation will host our first conference for the year. It will focus on the treatment of cancers of the upper digestive tract. I look forward to welcoming our guest speakers and attendees, who will share their insights and techniques in the ongoing fight against such diseases. Knowledge sharing is vital to our profession and this magazine is one of the means by which we can facilitate this. I commend this issue to you. n Editor-in-chief Marek Durlik, Warsaw Deputy Editor Irena Walecka, Warsaw Editor (English) Jansson Antmann Associate Editors Jarosław Buczek Magdalena Lewandowska Małgorzata Machaj Adam J. Sybilski Editorial board Piotr Andziak, Warsaw Maria Barcikowska, Warsaw Anna Czarnoba, Warsaw Andrzej Deptała, Warsaw Paweł Dobrzyński, Warsaw Marek Durlik, Warsaw Andrzej M. Fal, Warsaw Edward Franek, Warsaw Krzysztof Filczak, Warsaw Andrzej Gietka, Warsaw Robert Gil, Warsaw Stanisław Głuszek, Kielce Artur Jakimiuk, Warsaw Bogusław Kostkiewicz, Warsaw Dariusz Kosior, Warsaw Ireneusz Kotela, Warsaw Anna Nasierowska-Gutmajer, Warsaw Wojciech Rogowski, Warsaw Lidia Rudnicka, Warsaw Grażyna Rydzewska, Warsaw Andrzej Rydzewski, Warsaw Joanna Sempińska-Szewczyk, Warsaw Kazimierz Suwalski, Warsaw Piotr Suwalski, Warsaw Adam J. Sybilski, Warsaw Irena Walecka, Warsaw Jerzy Walecki, Warsaw Małgorzata Wisłowska, Warsaw Address CSK MSW Warszawa, 137 Wołoska St. tel ; fax redakcja@problemylekarskie.pl Publisher Central Clinical Hospital of the Ministry of the Interior Mossakowski Medical Research Centre, Polish Academy of Sciences 5
4 44 ROLA I MIEJSCE RADIOTERAPII W LECZENIU SKOJARZONYM NOWOTWORÓW GÓRNEGO ODCINKA UKŁADU POKARMOWEGO VALUE OF RADIOTHERAPY IN THE COMBINED TREATMENT OF UPPER DIGESTIVE TRACT TUMOURS Lucyna Kępka 8 INFLUENCE OF PREOPERATIVE VARIABLES THAT PROJECT OUTCOMES FOR ADULT PATIENTS WITH GLIOBLASTOMA MULTIFORME Kacper Kostyra, Kamil Adamczyk, Krzysztof Bojanowski, Katarzyna Dyttus-Cebulok, Bogusław Kostkiewicz 49 POWIKŁANIA ONKOLOGICZNE PO CHIRURGICZNYM LECZENIU NOWOTWORÓW GÓRNEGO ODCINKA PRZEWODU POKARMOWEGO UZASADNIENIE LECZENIA SKOJARZONEGO ONCOLOGICAL COMPLICATIONS AFTER SURGERY CANCER UPPER GASTROINTESTINAL TRACT EXPLANATORY COMBINATION THERAPY Wojciech Galla, Tomasz Kasprzyk, Zoran Stojčev 13 NEUROIMAGING AND SURGICAL TREATMENT OF LOW-GRADE GLIOMAS IN ADULTS Tomasz Tykocki, Bogusław Kostkiewicz 53 VERTEBRAL CANAL HAEMATOMAS REVEALED AFTER MILD TRAUMA IN CHILDREN 2 CASE REPORTS Marcin Tosik, Bogusław Kostkiewicz, Emilia Nowosławska 18 HEMODYNAMIC CHANGES AROUND CEREBRAL ARTERIOVENOUS MALFORMATION. PATHOPHYSIOLOGY AND TREATMENT Tomasz Tykocki, Magdalena Żychowska, Bogusław Kostkiewicz Basic diagnostic methods in dermatology 28 BASIC DIAGNOSTIC METHODS IN DERMATOLOGY PART I. DERMATOSCOPY AND VIDEODERMATOSCOPY Zuzanna Łagun, Marta Wieczorek, Elżbieta Szymańska, Irena Walecka 31 ROLA I MIEJSCE CHEMIOTERAPII W LECZENIU SKOJARZONYM NOWOTWORÓW GÓRNEGO ODCINKA UKŁADU POKARMOWEGO Andrzej Deptała 36 NOWOTWORY GÓRNEGO ODCINKA UKŁADU POKARMOWEGO: PROBLEM CHIRURGICZNY CZY ONKOLOGICZNY Andrzej W. Szawłowski Public Health creating a healthier nation Management 58 PREVENTION IS THE BEST MEDICINE INTERVIEW WITH DR N. MED BEATA MAŁECKA-LIBERA, SECRETARY OF STATE, GOVERNMENT PLENIPOTENTIARY FOR THE DRAFT PUBLIC HEALTH ACT. Jansson J. Antmann 38 OCENA PATOMORFOLOGICZNA NOWOTWORÓW GÓRNEGO ODCINKA PRZEWODU POKARMOWEGO Anna Nasierowska-Guttmejer 62 THE MUTUAL BENEFITS OF EMPLOYING PEOPLE WITH DISABILITIES Małgorzata Machaj, Marek Durlik, Maria Woźniak 6 7
5 INFLUENCE OF PREOPERATIVE VARIABLES THAT PROJECT OUTCOMES FOR ADULT PATIENTS WITH GLIOBLASTOMA MULTIFORME KACPER KOSTYRA¹, KAMIL ADAMCZYK², KRZYSZTOF BOJANOWSKI¹, KATARZYNA DYTTUS-CEBULOK³, BOGUSŁAW KOSTKIEWICZ¹ 1 Department of Neurosurgery, Central Clinical Hospital of the Ministry of the Interior, Warsaw, Poland 2 Student Scientific Circle of Dpartment of Neurosurgery, Central Clinical Hospital of the Ministry of the Interior, Warsaw, Poland 3 Cancer Center and Institute of Oncology in Warsaw Corresponding author: Kacper Kostyra Department of Neurosurgery, Central Clinical Hospital of the Ministry of the Interior, Warsaw, Poland Glioblastoma multiforme (GBM, WHO IV) is one of the most frequent and the most malignant primary astrocytic tumors of the brain (12 13% of all intracranial tumors) [17]. It develops from the diffuse fibrillary or gemistocytic astrocytoma, but in most caseses it is high grade glioma (HGG) from the beginning. Glioblastoma multiforme most frequently appears in the fifth decade of life. There are 2800 new cases of malignant, primary brain tumors in Poland annually and the majority of them are HGG [1]. GBM is an untreatable desease and despite many studies, including molecular treatment and advance diagnostic and surgical techniques, treatment results are still unsatisfactory. Mean survival of patients with glioblastoma multiforme is between 9 and 14,6 months and the quality of life systematicaly worsens [2-5]. It seems that there are a lot of factors which influence the prognosis, both preoperative: age, KPS, localisation, socioeconomic state and connected with treatment, molecular markers, genotype of the tumor, which are the subject of many actual studies [6-13, 19]. Abstract Object: The aim of this study, is the evaluation of the survival of patients with primary glioblastoma multiforme (GBM), according to the preoperative classification scale prepared by the M.D. K. Chaichana and coauthors and compare this results with data from the liturature. Methods: The authors have evaluated a group of 51 patients (26 women, 25 men), years of age, who underwent surgery for an intracranial primary (de novo) GBM between 2004 and 2005 in the Department of Neurosurgery at the Central Clinical Hospital of the Ministry of the Interior in Warsaw. All patients included in this study, underwent radio and chemioterapy after surgical treatment. Results Of the 51 patients in this study, 32 (82%) had died as of most recent follow-up (median time from surgery to death 9,9 months ± 7,7 months). During the study, there were 7 patients alive and the time of the observation varied from 10 months and 32 months (mean time was 16,29 months). The statistical analysis showed, that early prognostical factors which have the influence for the survival patients with glioblastoma multiforme are the Karnofsky performance scale (KPS) and the age more than 60. Keywords: preoperative variables, survival, glioblastoma multiforme There were a lot of attempts to find the prognostic factors which influence the survival of patients with glioblastoma multiforme. In most studiem, the most important preoperative factors that have been consistently shown to be associated with duration of survival are: age and functional status (KPS) [14-16]. Chaichana and coauthors, on the basis of their studies, have shown that despite treatment strategy, survival is influenced by: age, functional status and neurological deficits like aphasia and motoric deficits and tumor localisation [5]. The aim of this study is the evaluation of the survival of patients with primary glioblastoma multiforme (GBM) according to the preoperative classification scale prepared by the M.D. K. Chaichana and coauthors and to compare these results with data from the liturature. Methods The authors evaluated a group of 51 patients (26 women, 25 men), years of age, who underwent surgery for an intracranial primary (de novo) GBM between 2004 and 2005 in the Department of Neurosurgery at the Central Clinical Hospital of the Ministry of the Interior in Warsaw. The clinical, operative, and hospital course records of the patients were retrospectively reviewed. The information collected from neurosurgery and neurooncology clinical notes included patient demographic characteristics, functional status, comorbidities, presenting symptoms, results of neuroimaging, neurological function, tumor localisation, histopatological examination and adjuvant therapy. The pathology was determined by a senior neuropathologist in all cases, and the grading criteria were based on the WHO classification system. Patients of at least 18 years of age with a tissue-proven diagnosis of a primary supratentorial GBM (WHO Grade IV) were included in the study. Patients who had undergone prior resections, previous adjuvant therapy (chemotherapy or radiation therapy), infratentorial gliomas, and/or needle biopsies were excluded from the analysis. Patients with incomplete medical records lacking data with respect to clinical presentation, pre- and postoperative MR imaging, and/or adjuvant therapies were also excluded. This was done to create a more uniform patient population with similar tumor types, tumor location and treatment strategies. Information about follow up (time from surgery to death) was collected from the regional registry offices. 39 patients met the inclusion critria (20 women, 19 men) n the age of years (mean age 65,1 ± 15,2). The next stage of the study was the trial of preoperation factors which influence survival of patients with GBM and comparison with M.D. K. L. Chaichan and coauthors scale. One point was assigned to each factor independently associated with decreased survival of patients with GBM. One point each was assigned to patients over 60 years of age, KPS score 80, motor deficit, preoperative language deficit, and periventricular tumor location at the time of surgery. Patients therefore could have a prognostic score ranging from 0 to 5. Patients who received a score of 0 1, 2, 3, and 4 5 were assigned prognostic classes of 1, 2, 3, and 4, respectively. Summary data were analysed using: StatSoft, Inc. (2010). STATISTICA version Statistical Package R: Program Calc OpenOffice.org 3.3.0, Copyright 2000, 2010 Oracle and Calculator for confidence intervals of odds ratio, [18]. Results Median survival time 32 patients (82% of studied group) was 9,9 months ± 7,7months. During the study, there were 7 patients alive and the time of the observation varied from 10 months and 32 months (mean time was 16,29 months). Detailed data of the survival plot are shown in Figure 1. Statistical analysis proved that the early factors significantly associated with shortened survival were: an age over 60 and preoperative KPS score. No other clinical or imaging variables were found to be significantly associated with survival. In long rank analysis, a preoperative KPS score < 80 influences survival time and is statistically significant (p< 0,0065). Classes proposed by M. D. K. Chaichana and coauthors in the case of our study group were statistically significant, too (Chi square = 9,65; df = 3; p<0,05). Mean survival time of class 1 was 16,2 ± 10,6 ; class 2-10,2 ± 4,5; class 3-8,7 ±7,0 and class 4-6,3 ± 4,5 months. Despite this, on the basis of proportional hazards, the regression analysis by Cox showed that survival time of the study group was dependent upon age, which was the variable that hid the others (Table I). Motor deficits, aphasia and the tumor localisation were not statistically significant for survival time of patients (p<0,4; p<0,9; p<0,5). Discussion Preoperative clinical conditions of patients with glioblastoma multiforme are currently the subject of many studies. It seems that we can point out variable preoperative factors, which play a significant role in prognosis. Most authors are of the opinion, that the most important are: age, preoperative performance score by Karnofsky, tumor localisation and its size [14, 27, 28, 30]. Others tend to connect survival time with the neurological symptoms. Our study group comprised 39 patients with recognition of primary glioblastoma multiforme of the brain. At the same time 7 patients were alive during the study. The mean survival time was 9,9 months ± 7,7months. In our group, neurological deficits, sensory deficits, vertigo and headaches were the most popular initial symptoms. Incidence of occurrence was comperable with the references [5]. After conducting a multifactorial statistical analysis, taking into account factors which could have an impact on survival time of patients in our group, the most important were age and functional status according to the Karnofsky scale. Results have not shown a significant influence of tumor localisation and neurological symptoms like aphasia and motor deficits on prognosis in the case of patients with glioblastoma multiforme of the brain. A patient s age over 60 was directly connected with worsened prognosis. Similar results were revealed by other studies carried out to date [27, 28]. A senior age may have the impact of worsening a patient s ability to endure hardships connected with tumor symptoms, hospitalisation and treament. Moreover, like in references, elderly patients with glioblastoma multiforme may have a different molecular and genotypic profile of the tumor, which shows greater malignancy and treatment resistance. This feature can have the impact of worsening treatment results in elderly population of patients. Evaluating the functional status using Karnofsky scale is widely thought to be an authoritative method, specifying the proper state of oncology patients and together with age, it is believed to be a prognostic factor in the case of patients with glioblastoma multiforme [19-23]. Patients with poor preoperative functional status, tended to have decreased survival times, compared with patients in a good state of condition. In fact, among KPS score categories, KPS score 80 had the most significant association with poor survival and this score is comparable with literature. [27-29]. Patients with better functional status may be able to tolerate better neurological insults caused by the tumor, surgery, and/or adjuvant therapy. 8 9
6 Prawdopodobieństwo przeżycia 1,0 0,9 0,8 0,7 0,6 0,5 0,4 0,3 0,2 Funkcja przeżycia Kaplan-Meier a zgony pod obserwacją Prawdopodobieństwo przeżycia 1,0 0,9 0,8 0,7 0,6 0,5 0,4 0,3 0,2 Prawdopodobieństwo przeżycia Kaplan-Meier a zgony pod obserwacją Prawdopodobieństwo przeżycia 1,0 0,9 0,8 0,7 0,6 0,5 0,4 0,3 0,2 Prawdopodobieństwo przeżycia Kaplan-Meier a zgony pod obserwacją 0,1 0,1 0, Czas przeżycia Figure 1. KAPLAN-MEIER SURVIVAL PLOT FOR PATIENTS DIAGNOSED WITH GLIOBLASTOMA MULTIFORME Klasa 1 Klasa 2 Klasa 3 Klasa 4 KPS<80 pozostali Figure 2. KAPLAN-MEIER SURVIVAL PLOT FOR DIFFERENT PROGNOSTIC CLASSES Figure 3. KAPLAN-MEIER SURVIVAL PLOT FOR KARNOFSKY PERFORMANCE STATUS Table I. INFLUENCE FOR SOME PROGNOSTIC FACTORS ON SURVIVAL TIME Peoperative prognostic features Probability Relative risk Relative risk 95% lower 95% upper age 0, ,08 1,04 1,13 KPS score 80 0,20 2,37 0,63 8,92 motor deficit 0,99 1,00 0,33 3,00 language deficit 0,37 0,46 0,64 3,34 periventricular tumor localisation 0,52 1,30 0,59 2,85 It is extremely important to elaborate upon the effective preoperative classification scale for patients with intracranial GBM, which can affect the clinical outcome. It could be useful because there is no standard for guiding surgical and medical management, as there is for patients with arteriovenous malformations, intracranial hemorrhage or spinal injury [24-26]. As is well known, survival of patients with GBM is extremely unpredictable, with heterogeneous survival times. An effective classification system based on preoperative risk factors, could allow clinicians to prognosticate survival as well as possibly help guide surgical and medical strategies. This research confirms that, as has been described by many authors, preoperative factors like age and KPS, have an effect upon the prognosis of patients with intracranial GBM. Obtained own results clearly indicate that a patient s age is an irrefutable prognostic factor, which has the major influence upon survival, despite of other prognostic factors. Statistical analysis indicates that, as proposed by Chaichan and coauthors, the preoperative classification scale of patients with GBM had no essential influence upon the calculation of the survival time of our patients. This study, however, has some limitations. We have not analyzed the prognostic implication of molecular markers and genotypes that may also be linked to patient outcomes. In recent studies, patients with GBM and O6-methylguanine DNA methyltransferase (MGMT) promoter methylation had prolonged survival after temozolomide and radiation therapy, as compared with patients without this molecular marker. More recently, Parsons et al. [30] performed a genomic analysis in patients with GBM and found that patients with isocitrate dehydrogenase 1 (IDH1) mutations had prolonged survival times. These molecular markers and others may be associated with survival, but were not analyzed in this study. Furthermore, the majority of patients in this study did not undergo GTR and/or receive triple combinatorial adjuvant therapy (carmustine wafer, temozolomide, radiation). As a result, the relevance of this prognostic model may be altered in the context of those patients receiving the most aggressive of treatment regimens. Additionally, this study is inherently limited by its retrospective design, and, as a result, it is not appropriate to infer direct causal relationships. However, we tried to create a uniform patient population by utilizing strict inclusion and exclusion criteria, thus providing more relevant information for patients with primary intracranial GBM. We included only adult patients with primary GBM, since children and patients with secondary GBM may have tumors with different molecular and cytogenic profiles. In addition, we excluded patients with incomplete medical records and patients who had undergone prior resections, had received previous adjuvant therapies, had undergone needle biopsies, and had infratentorial tumors. Furthermore, we performed multivariate analyses and controlled for potential perioperative confounding variables. Given these statistical controls and a relatively precise outcome measure, we believe our findings offer useful insights into the management of patients with primary GBM. However, prospective studies are needed to provide better data to guide clinical decision-making. Table II. CLINICAL CHARACTERISTICS OF THE STUDY SAMPLE Preoperative patient s features Number of patients motor deficit 23 sensory deficit 20 language deficit 10 seizures 9 nausea/vomiting 9 headaches 8 visual deficit 5 confusion/memory loss 1 Conclusions The conducted study demonstrated that there is a statistical significant relationship between survival time of patients with intracranial GBM and the age at onset, and poor performance status expressed in the KPS score. Listed factors have an influence upon prognosis regardless of the applied therapy. There is a strong need to conduct further studies on factors which have an influence upon the prognosis of patients with intracranial glioblastoma multiforme and, above all, the definition of unequivocal, preoperative variables and a system which can classify patients to an appropriate risk group. n References 1. Mirimanoff R, Gorlia T, Mason W, et al. Radiotherapy and temozolomide for newly diagnosed glioblastoma: Recursive partitioning analysis of the EORTC 26981/22981-NCIC CE3 phase III randomized trial. J Clin Oncol 2006;24: Brada M, Sharpe G, Rajan B, et al. Modifying radical radiotherapy in high grade gliomas. Shortening the treatment time through acceleration. Int J Radiat Oncol Biol Phys, 1999; 43: Reni M, Cozzarini C, Ferreri A, et al. A retrospective analysis of postradiation chemotherapy in 133 patients with glioblastoma multiforme. Cancer Invest 2000;18: Shibamoto Y, Yamashita J, Takahashi M, et al. Supratentorial malignant glioma: an analysis of radiation therapy in 178 cases. Radiother Oncol 1990;18: Chaichana K, Parker S, Olivi A, Quiñones-Hinojosa A. A proposed classification system that projects outcomes based on preoperative variables for adult patients with glioblastoma multiforme. J Neurosurg 112: , Stummer W, Novotny A, Stepp H, et al. Fluorescenceguided resection of glioblastoma multiforme by using 5-aminolevulinic acid induced porphyrins: a prospective study of 52 consecutive patients. J Neurosurg 2000;93: Rainov NG, Dobberstein KU, Bahn H, et al. Prognosis factors in malignant glioma: Influence of the overexpression of oncogene and tumor-suppressor gene products on survival. J Neurooncol 1997;35: Shinoda J, Sakai N, Murase S, et al. Selection of eligible patients with supratentorial glioblastoma for gross total resection. J Neurooncol 2001;52: Mineo JF, Quintin-Roue I, Lucas B, et al. Glioblastomas: clinical study and prognosis factors. Neurochirurgie 2002;48: Lacroix M, Abi-Said D, Fourney DR, et al. A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection and survival. J Neurosurg 2001;95: Keles GE, Anderson B, Berger MS. The effect of extent of resection on time to tumor progression and survival in patients with glioblastoma multiforme of the cerebral hemisphere. Surg Neurol 1999;52: Stylli SS, Kaye AH, MacGregor L, et al. Photodynamic therapy of high grade glioma - long term survival. J Clin Neurosci 2005;12: Bussiere M, Hopman W, Day A, et al. Indicators of functional status for primary malignant brain tumor patients. Can J Neurol Sci 2005;32: Chang SM, Parney IF, McDermott M, Barker FG II, Schmidt MH, Huang W, et al: Perioperative complications and neurological outcomes of first and second craniotomies among patients enrolled in the Glioma Outcome Project. J Neurosurg 98: ,
7 MANAGEMENT 15. Lamborn KR, Chang SM, Prados MD: Prognostic factors for survival of patients with glioblastoma: recursive partitioning analysis. Neuro-oncol 6: , Laws ER, Parney IF, Huang W, Anderson F, Morris AM, Asher A, et al: Survival following surgery and prognostic factors for recently diagnosed malignant glioma: data from the Glioma Outcomes Project. J Neurosurg 99: , StatSoft, Inc. (2010). STATISTICA version Quigley M, Maroon J. The relationship between survival and the extent of the resection in patients with supratentorial malignant gliomas. Neurosurgery 1991;29: Devaux B, O Fallon J, Kelly P. Resection, biopsy, and survival in malignant glial neoplasma. A retrospective study of clinical parameters, therapy, and outcome. J Neurosurg 1993;78: Laws E, Parney I, Huang W, et al. Survival following surgery and prognostic factors for recently diagnosed malignant glioma: data from the Glioma Outcomes Project. J Neurosurg 2003;99: Green S, Byar D, Walker M, et al. Comparisons of carmustine, procarbazine, and high-dose methylprednisolone as additions to surgery and radiotherapy for the treatment of malignant glioma. Cancer Treat Rep 1983;67: Lamborn K, Chang S, Prados M. Prognostic factors for survival of patients with glioblastoma: recursive partitioning analysis. Neuro-oncol 2004;6: Spetzler RF, Martin NA: A proposed grading system for arteriovenous malformations. J Neurosurg 65: , Hemphill JC III, Bonovich DC, Besmertis L, Manley GT, Johnston SC: The ICH score: a simple, reliable grading scale for intracerebral hemorrhage. Stroke 32: , Harrop JS, Vaccaro AR, Hurlbert RJ, Wilsey JT, Baron EM, Shaffrey CI, et al: Intrarater and interrater reliability and validity in the assessment of the mechanism of injury and integrity of the posterior ligamentous complex: a novel injury severity scoring system for thoracolumbar injuries. Invited submission from the Joint Section Meeting On Disorders of the Spine and Peripheral Nerves, March J Neurosurg Spine 4: , Lamborn KR, Chang SM, Prados MD: Prognostic factors for survival of patients with glioblastoma: recursive partitioning analysis. Neuro-oncol 6: , Laws ER, Parney IF, Huang W, Anderson F, Morris AM, Asher A, et al: Survival following surgery and prognostic factors for recently diagnosed malignant glioma: data from the Glioma Outcomes Project. J Neurosurg 99: , Krex D, Klink B, Hartmann C, von Deimling A, Pietsch T, Simon M, et al: Long-term survival with glioblastoma multiforme. Brain 130: , Brem H, Piantadosi S, Burger PC, Walker M, Selker R, Vick NA, et al: Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet 345: , Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, et al.: An integrated genomic analysis of human glioblastoma multiforme. Science 321: , NEUROIMAGING AND SURGICAL TREATMENT OF LOW-GRADE GLIOMAS IN ADULTS TOMASZ TYKOCKI¹, BOGUSŁAW KOSTKIEWICZ² 1 Department of Neurosurgery, Institute of Psychiatry and Neurology, Warsaw, Poland 2 Department of Neurosurgery, Central Clinical Hospital of the Ministry of the Interior, Warsaw, Poland Correspondign author: dr Tomasz Tykocki ttomasz@mp.pl Department of Neurosurgery Institute of Psychiatry and Neurology in Warsaw Sobieskiego 9, Warszawa, Poland Introduction Low-grade gliomas (LGG) are defined on a neuropathological World Health Organization classification, but currently this term is commonly used in the clinical practice. LGG are characterized as benign brain tumors. They account for 15% of adult and 25% of pediatric primary brain tumors [1]. The incidence of LGG is estimated at approximately cases annually in USA. Males are more predisposed and biphasic age of distribution is observed with a first peak in the childhood (6 to12 years) and second between the third and fifth decades [2]. Along with the development of neuroradiology a substantially increased number of LGG is diagnosed. Routine application of MRI increases the sensitivity of the distinction of any kind of brain abnormalities, therefore LGG are detected at very early stage, still when the tumor is volumetrically very small. LGG grow very slowly and become asymptomatic for a long time. Clinical symptoms of raised intracranial pressure are very rare and the most often manifestation are seizures. Therefore, there is still much controversy about the first line of treatment in the early stage of LGG. Opinions vary, ranging from the fully conservative and only symptomatic treatment, through a stereotactic biopsy to aggressive surgical resection with additional chemo- and radiotherapy. Current recommendations from neurosurgical and oncological societies advise neurosurgical resection as the initial treatment. The principle of the operative methods is to achieve maximal tumor resection as possible and avoid neurological morbidity. To minimize postoperative neurological deficits, intraopertive MRI, brain mapping, motor and somatosensory evoked potentials are employed. However, Abstract Low grade gliomas (LGG) belong to a heterogeneous group of primary, benign brain tumors. LGG are slow-growing tumors, frequently asymptomatic for a long time. Seizures are often the first clinical symptom of LGG. Advanced neuroimaging techniques enable a more accurate diagnosis of LGG at earlier stages. There are still some controversies about the optimal management strategy of LGG. An initial stereotactic biopsy is recommended, as histological diagnosis influences subsequent decisions. Recent studies have shown that surgical resection should be the first line treatment. The aim of the surgical treatment of LGG is to maximize the extension of resection, while minimizing the risk of neurological deficits. As the majority of LLG infiltrate the eloquent area, the preservation of neurological functions is a neurosurgical challenge. The employment of intraoperative mapping and neurophysiological recordings has significantly decreased the rate of postoperative deficits. Total resection improves progression-free and overall survival rate. Seizure control and decreased malignant transformation are additional benefits. Low-dose, focal radiation is preferred in the postoperative period. Keywords: low grade gliomas, surgery, eloquent areas, mapping surgical treatment of LGG still becomes a great challenge, especially while balancing between the extension of resection corresponding with survival and the risk of new neurological deficits. Authors refer to the issue of extend of LGG resection on the survival trend in the treatment of LGG and analyzing current medical reports. Special remark is attracted to intraoperative techniques at resections of LGG at the eloquent areas. Neuroimaging of low-grade gliomas Adequate radiological investigation is crucial in early detection of LGG, especially taking into account their oligo- or asymptomatic course. The more precise radiological diagnosis, the earlier and more appropriate a neurooncological therapeutic strategy is applied. Computed tomography (CT) is employed as a first line imaging tool in neurological diagnosis. CT is especially useful in calcified tumors, what was estimated in 20% of astocytomas grade II, and 40% of oligodendrogliomas [1]. Contrast enhancement was observed only in 20% of oligostrogliomas. CT imaging may result in severe risk of misdiagnosis of LGG. Magnetic resonance imaging (MRI) provides better sensitivity for any brain tissue abnormalities and is preferred as an optimal approach for standardized radiological tool. LGG are visible as hypointensive on T1- and hyperintensive on T2-weighted imaging with moderate oedema. Although the sensitivity of MRI is very valuable, the specificity is rather questioned. Almost half of the suspicions on MRI as LGG were not confirmed in biopsies, where most of the cases revealed ana
8 plastic gliomas [4]. Contrast enhancement is not a reliable criterion for radiological diagnosis for LGG, while over 30% of LGG enhance and 15% of high grade gliomas (HGG) do not [5]. Surprisingly 60% of benign oligodendrgliomas demonstrate contrast enhancement on MRI [6]. Volume of contrast enhancement may serve as a predictor of malignant transformation. A threshold of 4 ml was determined as a radiological factor of malignant progression with poor survival prognostic [7]. FLAIR (fluid light attenuation inversion recovery) imaging is more proper technique for visualization of the margins of LGG, more precise than T1 and T2 sequences [8]. The annual growth rate of more than 8mm correlates with a median survival of 5,6 years and 15 years for a growth rate less than 8mm/year [9]. Studies with spin echo technique and threshold values of relative cerebral blood volume (rcbv) =1 differentiate grade II glioma from grade III with sensitivity- 92% and specificity- 33% [10]. Increased rcbv threshold to 2,18 allows for the distinguishing of LLG and HGG with sensitivity -100% and specificity -86% [11]. Angiogenesis is associated with poor clinical course, thus perfusion MRI could be employed as a tool for assessment of the clinical prognosis. A threshold of 1,75 (rcbv) may indicate for the time to progression (1 year >1,75 and 10 years <1,75) [12]. Perfusion MRI enables to navigate the best target area for biopsy of LGG. Higher rcbv corresponds with anaplastic areas and should be targeted, if detected, for the adequate histological diagnosis [13]. Differentiation of radionecrosis and tumor recurrence is not very convincing on MRI. More reliable data provides perfusion imaging with rcbv threshold <0,6 for necrosis and >2,6 for the tumor tissue [14]. LGG with their hypocellularity comparing to HGG, are preferentially well distinguished in diffusion-weighted MRI (DWI). DWI detects water diffusion directions using fractional anisotropy (FA) parameter. A threshold value of FA=1,88 is statistically significant for radiological gliomas grading [17]. The apparent diffusion coefficient (ADC) is sensitive to water component, brain blood barrier permeability and cellularity rate. Astrocytomas, in opposition to oligodendrogliomas, present significantly increased ADC [15,16]. Another radiological tool useful in differentiation gliomas grading is diffusion tensor imaging (DTI). DTI presents disruption of white matter tissue only in HGG [19]. Magnetic resonance spectroscopy (MRS) provides a measure of brain metabolism and image acquisition is composed of protons signal from each voxel. Since the gliomas are heterogeneous, the single voxel technique is not precise. LGG present very low Choline/Cr ratio and an increase in NAA, these criteria provide sensitivity 70-90% and specificity % for differentiation with HGG [20, 21, 22]. There are no clear evidences of the superiority of neither perfusion imaging nor MRS. The hyperglycolysis in oncotic cells, resulting form increased glucose transport and metabolism, is a predictor of an anaplasia. Position emission tomography (PET) with fluoro-d-glucose (FDG) has high accuracy for detecting of areas with malignancy. LGG represent tendency to normal or even lower FDG uptake rate, in contrast to HGG. Applying a threshold of 1,5 (tumor to white matter ratio) and 0,6 (tumor to gray matter ratio) a sensitivity of 95% and specificity of 77% were established [23]. FDG PET is a promising tool in the detection of radionecrosis and recurrent tumor. Radionecrosis appears as hypometabolic and tumor as normo/hypermetabolic areas. However, FDG is a specific marker only for metabolic value, the marker sensitive for cellular proliferation is methionine (Met). Methionine uptake increases with a higher grade of glioma. The sensitivity of LGG and HGG detection by Met PET is 61% and 97% respectively [24]. Functional magnetic resonance imaging and intraoperative mapping Over 80% of LGG are localized in eloquent areas (EA). These characteristics require precise preoperative functional imaging to avoid neurological deficits after tumor resection. Preoperative radiological investigation aims preferentially on localization of EA with respect to the surgical approach. Functional MRI (fmri) with the blood oxygen level dependent (BOLD) technique displays indirectly brain activity level by detection of local hemodynamic changes. fmri serves to provide functional neuroanatomy reliable enough for a neurosurgeon to perform non-complicated tumor resection. Verification of fmri results with direct electrical mapping (DEM) has overlapped in 100% for 20 mm and 87% for 10 mm accuracy area [25]. The concordance of fmri and DEM in intra-axial tumors was 25-63% (<1cm) and 29-75% (<2cm). For extra-axial tumors the overlapping results obtain in 33% and 42% of contradictory results [26, 27]. PET and fmri results of EA detection are similar, with the mean accuracy oscillating around 8 mm. PET results were concordant to DEM in 60-92%(<1cm) and 29-75%(>2cm). Functional imaging allows to determinate the hemispheric dominancy and language areas. The referable procedure is intracarotid test with apobarbital (IAP) known as Wada test. The correspondence of 90% between fmri and IAP in language function detection was observed [28]. Patients with left temporal lobe epilepsy are at higher risk of misdiagnosis. The sensitivity and specificity of fmri and DEM was 81% and 53% [29]. An evaluation of EA based only on anatomical morphometry without functional imaging or tests seems to be very risky, while there is a significant individual variability in cortical organization. Besides, the tumor mass and plasticity adaptive mechanism may affect the cerebral topography. The study of temporal lobe resection revealed that the distance form the temporal pole to the language area detected in intraoperative mapping varied from 3 to 9 cm. [31]. Awake craniotomy technique An awake craniotomy is performed under total intravenous anesthesia. Sedation is achieved with propofol (up to 100ug/kg/min) and ramifentanil (o,05 ug/kg/min). No miorelaxants are used during surgery to enable functional assessment. Extra analgetic bolous and lignocaine/marcaine injection are used for head holder fixation. Before dura opening, all the sedatives are discontinued. Some authors suggest infiltrating dura with lignocaine before opening, but this intervention may lead to cortical hyperpolarization and sever adverse events [32]. Awaking time varies from 20 to 50 minutes. The mean duration of DEM is 20 minutes. When seizure occurs, a cold Ringer s/saline irrigation or 1ml propofol bolous shoud be administrated. DEM is performed cortically and subcortically with bipolar hand-held stimulator, 1 mm electrodes with 5 mm space (Ojemann or Osiris Stimulator) with biphasic square wave pulses, each phase lasting 1 ms, in trains lasting 1msec for cortical and 1-2 msec for subcortical mapping at 60 Hz. The current range is from 1,5 ma to 6 ma [33]. Positive language response is detected when speech arrest appears. Mapping is performed with current range progression. The duration of the stimulation is 3-4 sec for language and 1-2 sec for motor mapping. Each area should be tested three times, with cortical volume of 5mm3 [32]. Speech arrest is positive when non-motor responses in the mouth and pharynx is observed. Motor mapping in non-dominant hemisphere is under general anesthesia with higher current intensity range 5-15mA. Motor response may vary from focal to multiple muscles action, especially when subcortical stimulation is applied [33, 34]. Motor evoked potentials recording are useful in assessment of motor pathways integrity and detection of brain ischemia. Survival and postoperative results The application of intraoperative brain mapping facilitates the identification and preservation of functional areas during resection. A margin of at least 1cm from EA is recommended during LGG removal. Sanai et al. have analyzed and reported neurological sequelae after LGG resection in EA. Initially, 8,4% (21/250) of patients worsened and 14% (35/250) had new speech deficits. After 6 months 92,8% (52/56) of those with early postoperative deterioration recovered to baseline and 7,1% (4/250) were with permanent deficits [35]. Authors concluded that patients with preoperative motor or language deficits are at higher risk of neurological progression postoperatively. This is probably related to the fact that functional fibres pass through the tumor mass. Additionally, positive intraoperative mapping response correlates with 3,8% of permanent neurological deficits and when no EA was found the risk decreases to 2%. Fifty percent of patients with LGG suffer from refractory epilepsy. After total or subtotal tumor resection seizure control rate is 80% and antiepileptic drugs are suppressed in every third patient. Seizure reappearance correlates with progression of tumor [36]. Along with the development of neurosurgical techniques, the ability to obtain better postoperative outcomes has significantly increased. Law et al. [37] have presented a retrospective analysis of 461 patients with LGG treated at the Mayo Clinic from 1915 through and 10-year survival rates were 35,5% and 22% respectively. After 10 to 15 years the survival trend of patients with LLG was comparable to the general population. Age at diagnosis was found to be a predictor of overall survival, which was different in various age groups, with poor prognosis for patients over 50 years old [5 year survival rate: (0-19 yrs) - 83%; (20-49yrs) -35%; ( 50yrs)-12%]. There was no statistical difference in survival between groups with or without postoperative radiotherapy ( 4000rads). Subtotal tumor resection correlates with 1,4 times higher risk of recurrence and 4,9 times higher mortality in comparison with total resection. The annual recurrence risk was calculated for 3,5% [38]. Duffau et al. have compared the results of two series of patients with DEM (n=122) and with no DEM (n=100). DEM group had a significantly lower rate of severe permanent deficits (6.5% vs 17%), a higher rate of total and subtotal resections (25% and 51% vs 6% and 37%, respectively), with survival significantly correlated with extend of resection [39]. Stereotactic biopsy is often recommended as an optimal, invasive diagnostic tool to provide histological diagnosis. The accuracy and reliability of biopsy has been raised in several studies. The concordance between the biopsy results with following radical resection of LGG on histopathological examination was ranged from 51% to 95% [40, 41, 42]. Generally, the postoperative results after LGG removal correlate with the extension of resection. The range of resection should be assessed on postoperative FLAIR imaging. When no abnormalities on FLAIR are found the resection was defined as total, residual tumor volume less than 10 ml as subtotal and more than 10mL as partial resection. No recurrence at 5 years was observed when total resection was performed, 16.7% had recurrence with subtotal and 38.5% with partial removal [48]. Nowadays, there is much controversy about the radiation treatment in LGG. Comparison of high-dose with low-dose radiation did not reveal higher overall survival for high-dose radiation [43, 44]. It was proven that radiotherapy might decrease cognitive functions and quality of life [45, 46]. However, patients with early postoperative radiotherapy had longer progression-free survival, but overall survival was not affected [47]. Focal radiation is preferred with total doses ranging from 45 to 60 Gy in fractions of up to 2 Gy. Conclusions The surgical treatment of LGG has become a therapeutic standard. The clinical results advocate the value of vast surgical resection as an independent factor of longer overall survival. Therefore, the operative treatment should be maximally focused on total resection. Although, radiological imaging provides helpful guidance, there is still a high risk of misdiagnosis. Therefore, an optimal protocol should include preoperative radiological diagnosis together with stereotactic biopsy. The majority of LGG are localized in eloquent brain areas and careful balancing between extended resection and the risk of new neurological deficits is required. The use of intraoperative mapping techniques with awake craniotomy has become a treatment of choice at LGG resection. There is no evidence of a higher survival rate after radiotherapy and additional postoperative radiotherapy is questionable. Focal radiotherapy might be recommended only in cases with refractory epilepsy. n References 1. Guthrie BL, Laws ER Jr. Supratentorial low-grade gliomas. Neurosurg Clin N Am Jan;1(1): Review. 2. McCormack BM, Miller DC, Budzilovich GN, et al. Treatment and survival of low-grade astrocytoma in adults Neurosurgery Oct;31(4):
9 MANAGEMENT 3. Lee YY, Van TP Intracranial oligodendrogliomas: imaging findings in 35 untreated cases. Am J Roentgenol. 1989; 152(2): Kondziolka D, Lunsford LD, Martinez AJ Unreliability of contemporary neurodiagnostic imaging in evaluating suspected adult supratentorial (low-grade) astrocytoma. J Neurosurg. 1993; 79(4): Al-Okaili RN, Krejza J, Woo JH et al. Intraaxial brain masses: MR imaging-based diagnostic strategy--initial experience. Radiology May;243(2): Jenkinson MD, du Plessis DG, Smith TS, Joyce KA, Warnke PC, Walker C. Histological growth patterns and genotype in oligodendroglial tumours: correlation with MRI features. Brain Jul;129(Pt 7): Tofts PS, Benton CE, Weil RS et al. Quantitative analysis of whole-tumor Gd enhancement histograms predicts malignant transformation in low-grade gliomas. Magn Reson Imaging Jan;25(1): Price SJ, Jena R, Burnet NG, et al. Improved delineation of glioma margins and regions of infiltration with the use of diffusion tensor imaging: an image-guided biopsy study. AJNR Am J Neuroradiol Oct;27(9): Pallud J, Mandonnet E, Duffau H, et al. Prognostic value of initial magnetic resonance imaging growth rates for World Health Organization grade II gliomas. Ann Neurol Sep;60(3): Weber MA, Zoubaa S, Schlieterk M, et al. Diagnostic performance of spectroscopic and perfusion MRI for distinction of brain tumors. Cancer Imaging (2006) 6, S32 S41, DOI: / Spampinato MV, Smith JK, Kwock L,et al. Cerebral blood volume measurements and proton MR spectroscopy in grading of oligodendroglial tumors. AJR Am J Roentgenol Jan;188(1): Law M, Young RJ, Babb JS, et al. Gliomas: predicting time to progression or survival with cerebral blood volume measurements at dynamic susceptibility-weighted contrast-enhanced perfusion MR imaging. Radiology May; 247 (2): Maia AC Jr, Malheiros SM, da Rocha AJ, et al. Stereotactic biopsy guidance in adults with supratentorial nonenhancing gliomas: role of perfusion-weighted magnetic resonance imaging. J Neurosurg Dec;101 (6): Sugahara T, Korogi Y, Tomiguchi S, et al. Posttherapeutic intraaxial brain tumor: the value of perfusion-sensitive contrast-enhanced MR imaging for differentiating tumor recurrence from nonneoplastic contrast-enhancing tissue. AJNR Am J Neuroradiol May; 21 (5): Kono K, Inoue Y, Nakayama K, et al. The role of diffusion-weighted imaging in patients with brain tumors. AJNR Am J Neuroradiol Jun- Jul;22(6): Tozer DJ, Jäger HR, Danchaivijitr N et al. Apparent diffusion coefficient histograms may predict low-grade glioma subtype. NMR Biomed Feb;20(1): Inoue T, Ogasawara K, Beppu T, et al. Diffusion tensor imaging for preoperative evaluation of tumor grade in gliomas. Clin Neurol Neurosurg Apr;107(3): Goebell E, Paustenbach S, Vaeterlein O, et al. Lowgrade and anaplastic gliomas: differences in architecture evaluated with diffusion-tensor MR imaging. Radiology Apr;239(1): Price SJ, Burnet NG, Donovan T, et al. Diffusion tensor imaging of brain tumours at 3T: a potential tool for assessing white matter tract invasion? Clin Radiol Jun;58(6): Law M, Yang S, Wang H, et al. Glioma grading: sensitivity, specificity, and predictive values of perfusion MR imaging and proton MR spectroscopic imaging compared with conventional MR imaging. AJNR Am J Neuroradiol Nov-Dec;24(10): Murphy M, Loosemore A, Clifton AG, et al. The contribution of proton magnetic resonance spectroscopy (1HMRS) to clinical brain tumour diagnosis., Br J Neurosurg Aug;16(4): Negendank WG, Sauter R, Brown TR et al. Proton magnetic resonance spectroscopy in patients with glial tumors: a multicenter study,. J Neurosurg Mar;84(3): Delbeke D, Meyerowitz C, Lapidus RL, et al. Optimal cutoff levels of F-18 fluorodeoxyglucose uptake in the differentiation of low-grade from highgrade brain tumors with PET. Radiology Apr;195(1): Chao ST, Suh JH, Raja S, et al. The sensitivity and specificity of FDG PET in distinguishing recurrent brain tumor from radionecrosis in patients treated with stereotactic radiosurgery. Int J Cancer Jun 20;96(3): Yetkin FZ, Mueller WM, Morris GL, McAuliffe TL, Ulmer JL, Cox RW, Daniels DL, Haughton VM. Functional MR activation correlated with intraoperative cortical mapping. AJNR Am J Neuroradiol. 1997;18: Reinges MH, Krings T, Meyer PT, et al. Preoperative mapping of cortical motor function: prospective comparison of functional magnetic resonance imaging and [15O]- H2O-positron emission tomography in the same coordinate system. Nucl Med Commun. 2004;.25: Krings T, Schreckenberger M, Rohde V, et al. Metabolic and electrophysiological validationof functional MRI. J Neurol Neurosurg Psychiatry. 2001; 71: Woermann FG, Jokeit H, Luerding R et al. Language lateralization by Wada test and fmri in 100 patients with epilepsy. Neurology. 2003; 61: Roux FE, Boulanouar K, Ranjeva JP, et al. Usefulness of motor functional MRI correlated to cortical mapping in Rolandic low-grade astrocytomas. Acta Neurochir (Wien). 1999; 141: Ojemann GA. Individual variability in cortical localization of language. J Neurosurg Feb; 50 (2): Davies KG, Maxwell RE, Jennum P et al. Language function following subdural grid-directed temporal lobectomy. Acta Neurol Scand Sep;90(3): Szelényi A, Bello L, Duffau H, et al. Intraoperative electrical stimulation in awake craniotomy: methodological aspects of current practice. Workgroup for Intraoperative Management in Low-Grade Glioma Surgery within the European Low-Grade Glioma Network. Neurosurg Focus Feb;28(2):E7. Review. 33. Sanai N, Berger MS Operative techniques for gliomas and the value of extent of resection. Neurotherapeutics Jul;6(3): Review. 34. Duffau H. Lessons from brain mapping in surgery for low-grade glioma: insights into associations between tumour and brain plasticity. Lancet Neurol Aug;4(8): Review. 35. Sanai N, Mirzadeh Z, Berger MS. Functional outcome after language mapping for glioma resection. N Engl J Med Jan 3;358(1): Branco DM, Coelho TM, Branco BM, et al. Functional variability of the human cortical motor map: electrical stimulation findings in perirolandic epilepsy surgery. J Clin Neurophysiol. 2003; 20: Laws ER Jr., Taylor WF, Clifton MB. Neurosurgical management of low-grade astrocytoma of the cerebral hemispheres. J Neurosurg. DOI: / jns @sup issue Claus EB, Horlacher A, Hsu L et al. Survival rates in patients with low-grade glioma after intraoperative magnetic resonance image guidance. Cancer Mar 15;103(6): Duffau H, Lopes M, Arthuis F et al. Contribution of intraoperative electrical stimulations in surgery of low grade gliomas: a comparative study between two series without ( ) and with ( ) functional mapping in the same institution. J Neurol Neurosurg Psychiatry Jun;76(6): Jackson RJ et al. (2001) Limitations of stereotactic biopsy in the initial management of gliomas. Neuro Oncol 3: Yu X et al. (2000) Stereotactic biopsy for intracranial space-occupying lesions: clinical analysis of 550 cases. Stereotact Funct Neurosurg 75: Aker FV et al. (2005) Accuracy and diagnostic yield of stereotactic biopsy in the diagnosis of brain masses: comparison of results of biopsy and resected surgical specimens. Neuropathology 25: Karim AB, Maat B, Hatlevoll R, et al. A randomized trial on dose-response in radiation therapy of lowgrade cerebral glioma: European Organization for Research and Treatment of Cancer (EORTC) Study Int J Radiat Oncol Biol Phys 1996;36: Shaw E, Arusell R, Scheithauer B, et al. Prospective randomized trial of low- versus high-dose radiation therapy in adults with supratentorial low-grade glioma: initial report of a North Central Cancer Treatment Group/Radiation Therapy Oncology Group/ Eastern Cooperative Oncology Group study. J Clin Oncol 2002; 20: Kiebert GM, Curran D, Aaronson NK, Bolla M, Menten J, Rutten EH, et al. Quality of life after radiation therapy of cerebral low-grade gliomas of the adult: results of a randomised phase III trial on dose response (EO- RTC trial 22844). EORTC Radiotherapy Co-operative Group. Eur J Cancer 1998; 34: Taphoorn MJ, Klein M. Cognitive deficits in adult patients with brain tumours. Lancet Neurol 2004; 3: Karim AB, Afra D, Cornu P, Bleehan N, Schraub S, De Witte O, et al. Randomized trial on the efficacy of radiotherapy for cerebral low-grade glioma in the adult: European Organization for Research and Treatment of Cancer Study with the Medical Research Council study BRO4: an interim analysis. Int J Radiat Oncol Biol Phys 2002; 52: Bello L, Gallucci M, Fava M et al. Intraoperative subcortical language tract mapping guides surgical removal of gliomas involving speech areas. Neurosurgery Jan;60(1):
10 HEMODYNAMIC CHANGES AROUND CEREBRAL ARTERIOVENOUS MALFORMATION. PATHOPHYSIOLOGY AND TREATMENT TOMASZ TYKOCKI¹, MAGDALENA ŻYCHOWSKA², BOGUSŁAW KOSTKIEWICZ³ 1 Department of Neurosurgery, Institute of Psychiatry and Neurology in Warsaw 2 I Faculty of, Warsaw Medical University 3 Department of Neurosurgery, Central Clinical Hospital of the Ministry of the Interior, Warsaw, Poland Corresponding author: Tomasz Tykocki ttomasz@mp.pl Department of Neurosurgery Institute of Psychiatry and Neurology in Warsaw Sobieskiego 9, Warszawa Poland Cerebral arteriovenous malformations (AVMs) are abnormal direct connections between arteries and veins of varying caliber without capillaries as an interposing element [62]. AVMs focus attention of neurologists and neurosurgeons because of their sophisticated nature and pathophysiological effect exerted on the hemodynamics of cerebral blood flow (CBF). The concept of vascular steal has gained numerous advocates and is the best proof that AVMs are not separate independent lesions but are part of brain circulation and together with other brain vessels should be treated as a whole. Understanding sophisticated nature of AVMs is required before choosing an optimal therapeutic method conservative management or invasive treatment, taking simultaneously into consideration hemodynamic consequences of AVM excision such as cerebral edema or hemorrhage to the adjacent parts of the brain. These hemodynamic disturbances seem to be grounded either in occlusive hyperemia as a consequence of venous obstruction or impaired autoregulatory mechanism leading to the speculated phenomenon of normal perfusion pressure breakthrough (NPPB) [99]. Altered autoregulation in the region of AVMs may contribute to the hyperperfusion after the obliteration or excision of large high-flow AVMs. Prediction of hemodynamic complications after AVM excision and early detection of NPPB Abstract The management of cerebral arteriovenous malformations (AVMs) is amongst the most advanced and challenging of neurosurgical procedures. The main purpose of AVMs treatment is the extirpation of the lesion as it is the only way to eliminate the risk of hemorrhage. With the progress of treatment modalities, the question has arisen as to which modality should be applied in a particular patient and whether the risk associated with the proposed treatment does not outweigh the risk of hemorrhage resulting from AVM natural history. The first step to answer the question has been to analyze the hemodynamic properties of the nidus and factors influencing the intranidal pressure in particular. Electrical models and computer simulations as well as animal models have contributed to better knowledge of baseline values including the distribution of flow, pressure and resistance within consecutive parts of the AVM. The next step has been to assess the effect of AVM resection or occlusion on hemodynamics in the neighborhood of the lesion and cerebral blood flow (CBF). Normal perfusion pressure breakthrough and occlusive hyperemia after microsurgical resection, hemorrhage or ischemic events are controversial complications but should be treated as real threats to optimize the choice of treatment modality. The aim of this review is to present pathophysiological basics of hemodynamic disturbances evoked by the presence of arteriovenous malformation and discuss hemodynamic implications of interfering within its angioarchitecture by means of three commonly used treatment modalities: microsurgery, endovascular embolization and radiosurgery. Keywords: arteriovenous malformations, hemodynamics, radiosurgery, microsurgery, embolization initial signs enables proper postoperative care [28]. The aim of this paper is to present distinctive hemodynamic features of AVMs, describe the way AVMs influence CBF and finally, emphasize hemodynamic disturbances after AVMs treatment with microsurgery, embolization or radiosurgery. The history of AVMs treatment The first historical information about any vascular malformation comes from the Papyrus Ebers, written in Egyptioan heratic writing dated on 1550 BC [107]. This historical manuscript contains descriptions of hemorrhoids, skin tumors, varicose veins and aneurysms. Another primary reports about vascular abnormalities could be found in Hippokrates s, Galen s, Celsus s, Aetius s, Avicenna s papers [107]. The breakthrough of understanding of cerebral circulation were made by Malpighi [56] (1661) who discovered capillaries, what was soon widely analysed by Harvey [32] (1628) and Willis (1664). William Hunter [37] (1757) was the first who presented the anatomical and hemodynamic features of arteriovenous malformation. He introduced the term of anastomosis contributing with previous description of erectile tumor. In the XIX century another over 20 terms referring to cerebral malformations were used. Rokitansky [83] concluded the neoplastic origin of malformations. This wrong hypothesis was supported by Luschka [55] who in fact, provided the description of two types of cerebral malformations: teleangiectases and cavernous tumors. The milestones for modern understanding of complex nature of AVMs were the papers written by Virchov [8]. They provided not only the anatomical issues but focus rather on pathophysiology and hemodynamics of AVMs. The first clinical diagnosis of a cerebral AVMs was probably made by Hoffmann [35] (1898), but the effective medical treatment still has been a real challenge. The first complete excision of a cerebral AVM was performed in 1889 by the famous French surgeon Jules Emile Pean [107] and in 1932 Olivercona [7] performed first successful removal of AVMs. He operated on a 37-year-old man with left cerebellar AVM. Cushing [17] and Dandy [19] published their AVMs operative experiences in 14 and 15 cases respectively in They reported various intraoperative troubles, especially those resulting from massive hemorrhage and probably too aggressive neurosurgical approach. Besides, those pioneers procedures couldn t be supported by angiography or other sophisticated brain scans. Both, the lack of knowledge on the morphology of AVMs or its size, location and number of draining vessels caused, that most of the operations had a great risk of morbidity and mortality. However, the introduction of cerebral angiography by Portuguese neurologist Moniz [64] in 1927 together with the improvements in the quality of angiograms has opened up new dimensions in the study of the morphological and hemodynamic aspects of AVMs. Two years later, Dott [21] presented the angiographic descriptions of AVMs for the first time. Therefore, in the 50s and 60s of last century the mortality ranged form o to 10 % after AVMs resections. Since then much more attention was paid to hemodynamic changes after operations, Murphy [68] was the first who reported the cerebral steal phenomenon. The continuing technological progress and the introduction of operating microscope, bipolar coagulation, microinstruments and neuroanesthetic technique improved postoperative clinical results. Spetzler and Martin [90] presented the correlation of the surgical results to size, location, relationship to eloquent area and venous drainage pattern. The further therapeutic innovations brought the development of non-surgical procedures. It was in 1967 when Lars Leksell [48] invented the gamma knife at Karolinska Institute in Sweden. The first report of an embolization of a traumatic carotico-cavernous fistula by Brooks [8] was in 1930 and endovascular embolization of cerebral AVMS was first described by Leussenhop and Spence in 1960 [54]. Pathophysiology Distinctive features of AVM hemodynamics are of particular interest to neurosurgeons due to their influence on the risk of hemorrhage and association with complications of various treatment modalities. The blood flow through sophisticated AVM angioarchitecture can be estimated using Hagen-Poiseuille formula (Q= P/R, in which Q is the flow, P pressure gradient, R-resistance) when we take into consideration that blood is a non-newtonian fluid, i.e. the blood viscosity depends on the shear rate and flow velocity, vessels are not rigid pipes but viscoelastic tapered tubes Clinical and the blood flow is not constant but pulsatile [44]. However, the Hagen-Poiseuile equation is applicable to vessels of diameter of at least 100µm [26]. As the diameter of feeding arteries tends to be larger, with the mean value of 800µm [97], the formula can be applied to AVM feeders and is the introduction to further calculations in artificially created models of AVM hemodynamics. The blood flow is inversely proportional to vascular resistance (R), which in turn can be expressed as R=8L η/ πr4 (where L is the length of the vessel, η blood viscosity, r inner radius of the vessel). Substituting this equation into Hagen-Poisseuille formula reveals that Q=π P r4/8l η. Assuming the flow to be proportional to the average blood flow velocity and vessel cross section (Q=v πr4) and substituting into the previous equation, we get v= P r4/8lη. Due to the lack of capillary bed in AVMs, blood flows directly to dilated low-resistance veins, which in turn leads to decrease in AVM resistance in relation to other cerebral vessels [5]. Flow resistance and resistive index (RI, systolic velocity minus diastolic velocity over systolic velocity) calculated from spectral Doppler waveforms may help distinguish between abnormal, low-resistant AVM vessels and normal en passant brain vessels during surgical resection of AVM, in particular in case of small lesions and residuals [18]. While pressures in feeding arteries and draining vein system can be measured directly using an intracranial microcatherer [22] during superselective angiography and mean values are reported to be 70mmHg in feeding arteries,18mmhg in draining veins of low-flow AVMs. In high-flow AVMs these values were 40mmHg in feeding arteries and 15mmHg in draining veins of [71], The intranidal pressure has been of particular interest and its contribution to hemorrhage, the most frequent AVM clinical presentation, has been studied thoroughly [67, 24, 29]. According to Doung et al. [22], high feeding artery pressure and deep venous drainage leading to outflow restriction are independent risk factors of hemorrhage, whereas in the same study nidus size, location and the presence of arterial aneurysms were found to be non-independent risk factors. Sorimachi et al. [88] demonstrated that feeding artery pressure tends to be lower in feeders with terminal divided branches (probably supporting separate AVM compartments) than in case of feeders with single branches terminating in the AVM. When the feeder has a transient branch to the eloquent part of the brain (supplying normal, relatively high-resistance brain tissue), the pressure in the feeder is found to be significantly higher. One way to confirm the speculated intranidal hemodynamics is to create electrical models of AVMs. Such models are based on the analogy between electric circuit and vessel network. Current is the analog of blood flow, voltage (the potential difference across two points) is compared to pressure (as pressure difference between two ends of the vessel causes blood flow), resistors in such artificially created circuits are equivalents of vessel resistance. The analogy between Ohm s law ( I=V/R in which I is the current, V-voltage, R-resistance) and Hagen-Poiseuille formula can be drawn. The disadvantage of such models is the simplicity of their architecture (often simulating only one feeding artery and draining vein or containing no compartments, whereas real, especially large high-flow AVMs, are usually either structur
11 MANAGEMENT ally or functionally compartmentalized lesions supported by 3-4 feeding arteries), the models don t provide for blood viscosity, shear stress, pulsatile blood flow, vessel distensibility and autoregulation [27]. A biomathematical model based on electrical network analysis, created by Hademonos et al. [31], determines intranidal hemodynamics with particular consideration of the differences between the plexiform and fistulous part of AVM. The mean intranidal flow was found to be 31,3ml/min with mean flow velocity of 66,4cm/s for the plexiform part and 617,6ml/min with mean flow velocity of 387,8cm/s for the fistulous part. The mean shear stress was 187,7 dyne/cm2 for the plexiform part and 463,5dyne/ cm2 for the fistulous part. Guglielmi et al.[27] have created two electrical models of low-flow (105ml/min) and highflow (550ml/min) AVMs to define hemodynamic conditions, including pressure and blood flow, within the successive parts of the AVM i.e. the arterial, arteriolar, arteriolar-venular transition, venular and venous parts of the lesion and assess the hemodynamic changes after endovascular embolization, surgical extirpation and bypass creation. The model revealed that in large, high-flow AVM, in relation to lower resistance of the nidus when compared to small AVM, the pressure regimen through the nidus is lower (especially when pressures on the arterial side of large and small AVMs are compared, 40 and 70mmHg respectively), the pressure in the arteriolar part of the nidus is lower (25,5 vs 44mmHg in small AVM), the flow through the venular part of large AVM is higher (48ml/min) and the pressure in the venular part is lower (18,5mmHg) than in small AVM (26ml/min and 24,5mmHg, respectively). The study confirmed large blood flow through the fistulous part (365ml/min) in comparison with the plexiform part (190ml/min) of the AVM nidus. Although one might expect to find a correlation between low feeding mean arterial pressure (FMAP) and high draining vein pressure (DVP) in high-flow fistulas, recent studies do not confirm such association. According to Young et al. [108], higher FMAP is directly related to higher DVP. In highflow AVMs the transnidal pressure (the difference between FMAP and DVP) seems to be lower, which results in decreased susceptibility to spontaneous vessel rupture. The resistance of the feeding arteries in large AVM was calculated to be lower than in small AVM (36Ω vs 240Ω) due to larger cross section of feeding arteries of large AVM. Due to larger cross section of draining veins of large AVM, the resistance of the draining veins was lower than in small AVM (13Ω vs 95Ω). An interesting finding is lower resistance of brain vessels in case of large, high-flow AVM than in small AVM (149 Ω vs 249 Ω). Brain vessels tend to dilate in order to maintain sufficient blood supply to the brain tissue. When the pressure drops beyond autoregulatory capabilities, the brain becomes hypoperfused and neurological deficits tend to appear. Large high-flow AVMs contribute to redistribution of blood flow, more blood is directed to the low-resistant lesion, which in turn leads to decrease in perfusion pressure of the brain-nutrifying vessels and the vascular steal phenomenon may occur if autoregulation range stays unchanged [29, 86]. However, symptoms ascribed to steal are a rare finding in AVM patients thanks to adaptive autoregulatory displacement [108]. The steal phenomenon is claimed to be responsible for progressive neurological deficits observed in approx 15% of patients with AVMs [12]. Sekhon et al. [85] have demonstrated by creating an arteriovenous fistula model in the rat that chronic non-infarctional ischaemia with the 25-50% reduction in CBF leads to the impairment of neuronal function. On the basis of analysis of 65 patients with AVM, Marks et al. [57] have defined morphological features associated with the risk of vascular steal, including large nidus (mean volume of 105cm3 in patients with the presentation of vascular steal, comparing with mean volume of 19,5cm3 in patients without such presentation), angiomatous change and peripheral venous drainage. Many scientists, on the other hand, diminish the importance of vascular steal. In a study carried out by Mast et al. [60] 11 out of 152 patients with AVM presented nonprogressive focal neurological deficits while only 2 patients suffered from progressive deficits. Patients with hemorrhage prior to neurological deficits were excluded from the study. The authors compared pressure and mean flow velocities in feeding arteries and found no difference between patients with versus without deficits. Another approach to shed light on the incompletely understood issue of AVM-related hemodynamic disturbances is to create such lesions in animals. This approach is grounded in a theory of AVM pathogenesis, assuming that morphological changes leading to the nidus formation occur secondary to increased blood flow and shear stress in a congenital arteriovenous fistula [66]. An affirmation of this theory can be effective attempts of the formation of human-like AVMs on the basis of surgically created arteriovenous fistula in animals. Wakhloo et al. [105] have proposed increased flow through the fistula and vessel remodeling to be responsible for reduction of muscular tone, vessel dilatation (mean diameter of 520µm after 6 months since fistula creation vs 320µm in control animals) and decreased resistance leading to the formation of high-flow AVM. Similarities between animal models and human AVMs were histopathologically and ultrastructurally verified and included ectasia of the model vessels, focal intimal hyperplasia, increased collagen deposition, disruption of the elastic lamina and the presence of filopodia on the luminal surface of endothelium [98]. Other important factors leading to hemodynamic perturbations and, therefore, suspected to be associated with the risk of rupture, are the presence of intranidal anerysms (or venous ectasias) and venous stenosis, considered by Krings et al. [45] to be focal weak points in AVM angioarchitecture. Aneurysms within feeding arteries or nidus as well as in remote vessels are frequent anomalies, estimated to be present in 10-58% of AVM patients [82] and may rupture before, during or immediately after AVM treatment [96]. Flow-related aneurysms tend to develop in sites of maximal shear stress, which are branching points of major feeding arteries. Therefore it is not surprising that they disappear after restoration of normal hemodynamic conditions of CBF [16]. Venous stenosis is supposed to be an excessive response of the venous wall to high flow and shear stress. Its correlation with the risk of rupture is disputable and probably depends on the selection bias of the patients. However, many studies confirm positive correlation with the risk of hemorrhage [63, 69, 103]. As intranidal aneurysms and venous stenosis are considered to be hazardous structures, Hirai et al. [34] have recommend focusing the treatment strategy on these lesions. In case of aneurysms both embolization and radiosurgery can be performed. In large AVMs with venous stenosis, embolization seems to be treatment of choice. Treatment The main aim of AVM treatment is to eliminate the risk of future hemorrhage by total occlusion or complete excision and preserve neurological function. The aim can be achieved after thorough clinical examination and radiological assessment by adjusting the treatment modality to patient s characteristics and AVM angioarchitecture in the way that the treatment-related morbidity and mortality didn t outweigh the risk of complication in the AVM natural history [73]. Microsurgery As observed by Guglielmi [27] after excision of lowflow AVMs the feeding artery pressure increases from 70 to 96mmHg with a simultaneous decrease of draining vein pressure from 18 to 6mmHg. In case of high-flow AVMs, the feeding artery pressure rise tends to be larger (from 40 to 87mmHg) with the draining vein pressure drop from 15 to 3mmHg. These changes result from removal of the lesion of low resistance to blood flow. In accordance with Poiseuille-Hagen formula, as the feeding arteries are permanently dilated and are unable to constrict immediately after AVM excision, the new pressure gradient must be reduced to maintain the same blood flow. This leads to a greater than normal blood pulsatility and may result in hemorrhage from a ruptured aneurysm or other site of vessel weakness [70]. After excision of high-flow AVM the autoregulatory mechanism has to intervene because large amounts of blood supplying previously the low-resistant lesion are now directed to dilated vessels of the adjacent brain [27]. Since years scientists have tried to answer the question whether the capillaries in the neighborhood of the nidus, adapted for chronic ischaemia are capable of autoregulation like capillaries of the distant to the AVM sites of the brain. If not, the risk of NPPB arises [4, 71, 91]. The phenomenon is due to the impairment of autoregulation, in particular in the shift of upper limit to the lower pressure value in cases of chronic brain hypoperfusion induced by the presence of a low-resistant lesion [38]. Paralyzed vessels adjacent to the AVM are unable to response to the restoration of normal perfusion pressure, which results in hyperemia and disruption of the capillary bed. NPPB occurs mainly in patients with large, high-flow AVMs, supplied by the branches of external carotid artery [91]. Hemorrhage during or after surgery is the most devastating complication. The larger the AVM, the higher the risk of hemorrhage. However, NPPB is only one explanation for postoperative hemorrhage and/or cerebral edema after AVM excision. A theory, introduced by al-rodhan [1] is based on studies that reduced blood flow through the draining veins with a simultaneous stagnant flow in the feeding arteries may result in intravenous clot formation. Obstruction of venous outflow leads to occlusive hyperemia. Distinctive features of AVM angioarchitecture, including (1) high-flow AVMs, (2) long and tortuous feeding arteries, (3) small number of draining veins, (4) pre-operative venous narrowing or occlusion and (5) the presence of vascular steal, are found to be associated with the elevated risk of occlusive hyperemia [1]. The grading scale comprised of nidus size, pattern of venous drainage and eloquence of the adjacent brain, proposed by Spetzler and Martin [90] in 1986, has been gold standard to assess the risk and predict the outcome of surgical intervention in AVM patients. Other grading systems, developed by Tamaki et al. [93], Hollerhage et al. [36], Pertuiset et al. [76] or Spears et al. [89] have not met with general approval. In 2010 Lawton et al. [46] have presented a grading scale, based on patient age, sort of clinical presentation (hemorrhagic or unhemorrhagic) and AVM diffuseness, as supplementary to Spetzler-Martin grading scale in order to increase the sensitivity, specificity (or both) of predicting the resection outcome. The authors based on statistically significant variabilities found to be independent risk factors of neurological complications after surgery (age, unhemorrhagic presentation, diffuse lesion). The aim of this simple scale is both to confirm the surgery risk estimated by means of Spetzler-Martin scale (when the outcome of the supplementary scale corresponds with the Spetzler-Martin outcome) and, which has more important clinical implications, to help to decide in doubtful cases of borderline outcome by Spetzler-Martin grading scale (with stress on Grade III subtypes). As Sanchez-Mejia [84] recommends, microsurgical resection should be done in case of radiated AVMs that are not completely obliterated after the 3-year latency period, but altered favorably for surgery, even in asymptomatic patients. Embolization Endovascular embolization is an important facilitation of surgery, aimed at reducing the AVM volume and flow and occlusion of deep, surgically inaccessible feeding arteries and intranidal fistulous compartments [101]. It is a part of multimodality treatment when combined with microsurgery or radiosurgery, but it may be also applied as the sole treatment method to achieve complete obliteration or as palliative treatment [73]. The data concerning postembolization mortality and morbidity vary significantly. Taylor et al. [94] highlight the fact that despite significant surgical facilitation measured in reduction of intraoperative blood loss and operative time, embolization is correlated with increased mortality and morbidity. According to the retrospective study carried out by the authors in 2004, 11% of patients died or suffered from a permanent neurological deficit after the embolization procedure. The data presented by Deruty et al. [20] revealed a 25%-complication rate (including minor deterioration, neurological deficits and death) after endovascular embolization, compared to minor complications (mainly minor deterioration) observed in 17% of surgically treated patients and 10% of irradiated patients. Such high postembolization complication rates may be only partially attributed to the fact that embolization is performed in sophisticated AVM cases as severe complications after embolization have been observed in low-grade AVMs as well. Therefore Deruty et al. [20] dissuade from performing embolization in low-grade AVMs and favour microsurgical 20 21
12 resection or irradiation in such cases. However, updates on the postembolization clinical outcome are more optimistic. A study conducted by Ledezma et al. [47] revealed 6,5% complication rate and 1,2% mortality rate. Haw et al. [33] reported 3,9% risk of death or permanent neurological deficit after embolization. The postembolization hemodynamic complications include both hemorrhage and ischemic events (50% each [20]), which may lead to transient or permanent neurological deficits. Ischemic events may result from occlusion of arterial branch supplying normal brain tissue or arterial dissection during endovascular access. Postembolization hemorrhage in the early period after the embolization, reported in 3-15% of patients and 1-2% of procedures [40] [77], is supposed to be a result of the increase of feeding artery pressure. Several factors, including (1) compact nidus, (2) large number of feeding arteries, (3) the presence of steal phenomenon, (4) preembolization venous stenosis or ectasia, (5) extensive venous embolization, have been postulated to predispose to early hemorrhage [77]. Hemorrhage in the late period occurs due to venous occlusion, being a result of slowed blood flow in the dilated veins and clot formation in the draining vein system, or the speculated phenomenon of NPPB. A separate issue is the periprocedural hemorrhage secondary to mechanical vessel perforation, AVM rupture or intranidal aneurysm rupture during catheterization [41]. The hemodynamic consequences of embolization depend on the magnitude of flow and the part of the AVM, which is occluded with the embolic agent. As the electrical models created by Gugliemi [27] demonstrate, the most desired effect is the occlusion of arterial feeders resulting in the reduction of blood pressure and flow through the nidus both in cases of low-flow and high-flow AVMs (with a 40% reduction of pressure and 50% reduction of flow through the nidus in small AVMs). However, if the embolic agent, after occlusion of the feeding arteries, does not penetrate to the shunt within the nidus, the risk of neoangiogenesis or reopening of the collateral circulation arises, which will make subsequent endovascular procedures nearly impossible [45]. On the other hand, occlusion of the draining vein system is an unwelcome result of embolization leading to a dramatic decrease of flow through the nidus and increase of pressure in the venular part of the nidus (in low-flow AVMs even by 284%) with high risk of postembolization hemorrhage [27]. Extreme caution is required during embolization of AVMs combined with venous stenosis as when even scant amount of embolic agent reaches the stenosed vein, the pressure within the nidus may increase significantly with severe hemodynamic consequences. Rupture may also be a result of occlusion of the fistulous compartment and the venular part of one of the channels of the plexiform compartment in high-flow AVMs, as the increase of flow and pressure within the nidus in such cases is noted to be 108% and 244%, respectively. However, Yuki et al. [110] demonstrated that embolization of the high-flow fistula, as a part of multimodality approach to cerebral AVMs with large fistulous compartments, was successful in 100% of patients, and the complications, if occurred, were associated with endovascular occlusion of other parts of the nidus. Single photon emission computed tomography (SPECT) enables measurement of regional cerebral blood flow (rcbf) in close proximity to the AVM and may show either postembolization disappearance of rcbf decrease, observed before the procedure, or increase of rcbf combined with hyperemic complications [6]. In large, high-flow AVMs the risk of NPPB seems lifelike, especially when one-staged complete excision is performed. Therefore, many authors recommend multimodality treatment composed of staged embolization and final surgical resection. The key issue is the choice of optimal interval between the embolization and surgery as too short intervals between the procedures may contribute to severe hyperemic complications and within long intervals the occluded vessels may recanalize or collateral circulation may develop. Chioffi et al. [11] observed higher risk of intraoperative complications including severe blood loss and periventricular hemorrhage as well as postoperative hemodynamic complications such as cerebral edema or intracerebral hematoma when the AVM mean flow velocity exceeded 120cm/s before the surgery. The issue that has been particularly under discussion is the choice of the optimal embolic agent considering the safety and effectiveness of the procedure. Embolic materials can be divided into two groups: solid agents, including polyvinyl alcohol particles (PVA), fibers, microcoils, microballoons and liquid agents including cyanoacrylate monomers e.g. I-butyl cyanoacrylate (IBCA) and N-butyl cyanoacrylate (NBCA), and polymer solutions e.g. ethylene vinyl alcohol [73]. The introduction of liquid agents revolutionized endovascular procedures and NBCA has been the most popular embolic material so far, with complete obliteration rate of approximately 10% [24], achieved mainly in small AVMs with a small number of feeding arteries [102], especially when the nidus is not compartmentalized or the fistulous compartment is dominant [100]. It has been postulated that the use of particle materials, as they induce slower obliteration, is associated with increased risk of hemorrhage due to slow but continuous increase of pressure within the nidus before complete obliteration [73]. Liquid agents allow the use of flow-directed catheters, which in turn eliminates the risk of mechanical perforation during the procedure and enables precise placement of the catheter in close proximity to the nidus, reducing the risk of ischemic events [106]. Ethylene vinyl alcohol copolymer (Onyx) is a new liquid agent enabling, at least theoretically, better penetration and simultaneous angiographic control [75]. However data concerning its effectiveness vary significantly, the complete obliteration rate ranging from 29%, reported by Tevah and Huete [95], to 0% in a study conducted by Jahan [42]. In 2010 a grading scale for AVM endovascular procedures, similar to the surgically applicable Spetzler-Martin grading scale, has been created by Feliciano et al. [23] However, the scale, composed of the number of feeding arteries, presence of fistulous compartments and eloquent location of the lesion, was based only on the analysis of outcome predictive factors described in literature and, as the authors mention, needs further examination. Embolization is performed when the nidus diameter is in excess of 3cm or it is supplied by deep feeding arteries with difficult surgical access [73]. In fear of too rapid redistribution of blood flow and subsequent hemorrhage and/or brain edema, staged embolization of the feeding arteries is recommended. Complete surgical excision should be performed within several days (1-2 weeks) after final embolization [73]. Radiosurgery The advantage of radiation in AVM treatment is its ability to cause endothelial damage and hyaline thickening, resulting in thrombosis and AVM obliteration [72]. Radiosurgical treatment is preferred in patients with unruptured, small (the efficiency of this modality is limited to lesions not greater than 3cm), deep AVMs, especially when the lesion is located in eloquent part of the brain (considered in such case to be treatment of choice) [73]. It is also a part of multimodality approach to treatment of giant AVMs associated with high surgical risk [73]. Radiosurgery prior to microsurgery facilitates the resection by (1) reducing the size of high-flow AVMs, (2) making the walls of the radiated arteries thicker and subsequently easier to occlude, (3) reducing the blood flow through the nidus and (4) inducing gliosis surrounding the lesion, which facilitates the dissection. As observed by Sanchez-Mejia [84], radiosurgery reduces the operative morbidity, graded according to the modified Rankin Scale (mrs). As the effect is not immediate and the latency period necessary to obtain obliteration is estimated to be approximately 2 years, one must contemplate the risk of hemorrhage within the latency period versus the capability to immediate AVM excision [10]. Numerous studies have been conducted to shed light on the risk of hemorrhage associated with the radiation-induced altered flow patterns within the latency period [15, 43, 53]. As the radiation induces thickening of the nidus vessel walls and subsequent fibrosis, one can expect the risk of hemorrhage within the latency period to be decreased. The data vary due to the sort of analysis and the annual risk of hemorrhage within the latency period is reported to be 1,5-3,7% [14, 87, 92]. In Karlsson s [43] study advanced age and low dose of radiation were found to be associated with higher risk of rupture. As a confirmation of Karlsson s observations may serve a biomathematical model of the influence of radiation on the risk of hemorrhage, developed by Massoud et al. [59] The authors stressed the elevated risk of rupture associated with partial and/or low-dose radiosurgery. When incomplete radiosurgery is performed, the resistance to outflow in occluded vessels increases and the blood is redistributed to unoccluded parts of the nidus, which results in generation of additional biomechanical stresses increasing the risk of rupture. Interestingly, nidus vessels in close relation to the feeding arteries and/or draining veins are the most susceptible to rupture [59]. Several authors highlight the risk of venous hypertension and occlusive hyperemia as a result of radiosurgically-induced venous occlusion prior to arterial occlusion. The subsequent increase of intranidal pressure may contribute to intracranial hemorrhage. According to Pollock [78], venous hypertension was present in 2 of 28 patients with symptomatic postradiosurgical imaging changes. Chapman et al. [11] described two cases of patients with delayed neurological deficits due to postradiosurgical venous outflow obstruction. Celix et al. [9] consider inflammatory response to irradiation as the most probable cause of venous thrombosis leading to hemorrhage in the early period following radiosurgery. Therefore, occlusive hyperemia, the term used primarily to explain pathophysiological basis for brain edema or hemorrhage after AVM surgical resection, may be responsible for hemodynamic complications after AVM radiosurgery, too. Other factors, including AVM size, the presence of aneurysms and hemorrhage within one year prior to radiosurgery, have been demonstrated to exert effect on the generation of hemorrhage after AVM radiosurgery [80, 49]. As demonstrated by Lo [51], the distribution of shear stress to the AVM shunt walls, which is directly related to the risk of rupture, is dependent on the mechanism of AVM obliteration. Two patterns of post-radiosurgical occlusion have been described in literature. The first one, less likely, referred to as random occlusion, signifies complete obliteration of only some of the nidus vessels and subsequent blood redistribution. The second mechanism of vessel occlusion is grounded in stepwise thickening of the vessel walls and narrowing of the lumen ( stepwise occlusion ) with eventual vessel occlusion [52]. The pattern of occlusion is probably much more complex in in vivo conditions than in theoretical considerations and requires allowing for dynamic phenomena within the nidus [59]. An incompletely understood phenomenon is the occurrence of hemorrhage after complete radiosurgical obliteration confirmed in angiography. Delayed hemorrhage was reported in 0,7-2,4% of patients [39, 58]. The speculated cause of such hemorrhage is recanalization of a small postradiosurgical thrombus, undetectable via neuroimaging. The suspected risk factors include young age, female sex and the presence of small AVM [61]. Reexamination of such patients after complete radiosurgical obliteration is recommended, as there is diminutive risk of AVM reappearance [50]. According to Sanchez-Mejia [84], radiosurgery is recommended for unruptured AVMs that are not favorable for microsurgical resection. As Spetzler-Martin grading scale does not correlate with patient outcome after AVM radiosurgery, Pollock and Flickinger [79] developed radiosurgery-based arteriovenous malformation score (RBAS). The authors formulated an equation to calculate the AVM score, associated with patient outcome after single irradiation. The AVM score can be defined in the following way: AVMscore=(0.1)(AVM volume in cm3) + (0,02)(patient age in years) + (0.3)(location of lesion: 0 if frontal or temporal; 1 if parietal, occipital, intraventricular, corpus callosum or cerebellar; 2 if basal ganglia, thalamic or brainstem). Lower AVM score (1 or less) is correlated with better clinical outcome. The grading scale, proposed in 2002, has been validated several times [3, 65, 81] and seems to be an accurate predictor of excellent outcome not only after single radiosurgery but also after retreatment and may help predict the clinical outcome after overall radiosurgical treatment [81]. Conclusions AVM are among those brain pathologies that have been for centuries of particular interest. Its complexity results from the angioarchitecture and hemodynamic properties. The essence of AVMs treatment relies on a multimodal ap
13 MANAGEMENT proach and commitment of a comprehensive therapeutic team. The development of radiosurgery, microsurgery and endovascular techniques enables to select of the best therapeutic method depending on the type of AVMs, as well as in specific cases, a combination of these methods. Large AVMs in the past, considered to be inoperable, can now be safely treated. In addition, apart from the significant difficulties arising from the exclusion of the AVM from cerebral circulation, equally important issue is to predict, control and prevention of hemodynamic consequences after surgery. Therefore, before proceeding with treatment, all matters relating to hemodynamic complications in any case should be examined. After a detailed analysis, an appropriate single or multimodal method of treatment should be selected, considering the risk of postoperative complications. The perfect course of treatment does not guarantee the avoidance of serious hemodynamic and functional impairment later on. NPPB, occlusive hyperemia, steel phenomenon are particularly dangerous and can cause severe clinical deterioration of patients. However, present clinical data confirms that AVMs could be safely treated with greatly reduced risk of serious complications compared with earlier results. References 1. Al.-Rodhan NR, Sundt TM Jr, Piepgras DG et al. (1993) Occlusive hyperemia: a theory for the hemodynamic complications following resection of intracerebral arteriovenous malformations. J Neurosurg 78(2): Al-Shahi R, Warlow C (2001) A systematic review of the frequency and prognosis of arteriovenous malformations of the brain in adults. Brain 124: Andrade-Souza YM, Zadeh G, Ramanti M et al (2005) Testing the radiosurgery-based arteriovenous malformation score and the modified Spetzler-Martin grading system to predict radiosurgical outcome. J Neurosurg 103(4): Barnett G, Little JR, Ebrahim ZY at a (1987) Cerebral circulation during arteriovenous malformation operation. Neurosurgery 20: Barrow D, Dawson R (1994) Surgical management of arteriovenous malformations in the region of the ventricular trigone. Neurosurgery 35: Batjer HH, Purdy PD, Giller CA at al (1989) Evidence of redistribution of cerebral blood flow during treatment for an intracranial arteriovenous malformation. Neurosurgery 25: Bergstrand H, Olivecrona H, Tonnis W (1936) GefaBmiGbildungen und Gefafigeschwulste des Gehirns. Thieme. Leipzig, p Brooks B. (1968) The treatment of traumatic arteriovenous fistula. South Med J 1930;23: Celix JM, Douglas JG, Haynor D et al (2009) Thrombosis and hemorrhage in the acute period following Gamma Knife surgery for arteriovenous malformation. Case report. J Neurosurg 111(1): Chaddad-Neto F, Joaquim AF, dos Santos MJ et al (2008) Microsurgical approach of arteriovenous malformations in the central lobule. Arq Neuro-Psiquiatr 66(4): Chapman PH, Ogilvy CS, Loeffler JS (2004) The relationship between occlusive hyperemia and complications associated with the radiosurgical treatment of arteriovenous malformations: report of two cases. Neurosurgery 55(1): Chioffi F, Pasqualin A, Beltramello A et al (1992) Hemodynamic effects of preoperative embolization in cerebral arteriovenous malformations: evaluation with transcranial Doppler sonography. Neurosurgery 31(5): Choi JH, Mast H, Hartmann A, Marshall RS, Pile-Spellman J, Mohr JP, Stapf C (2009) Clinical and morphological determinants of focal neurological deficits in patients with unruptured brain arteriovenous malformation. J Neurol Sci 287(1-2): Coffey RJ, Nichols DA, Shaw EG (1995) Stereotactic radiosurgical treatment of cerebral arteriovenous malformations. Gamma Unit Radiosurgry Study Group. Mayo Clin Proc 70(3): Colombo F, Pozza F, Chierego G, Casentini L, De Luca G, Francescon P. (1994) Linear accelerator radiosurgery of cerebral arteriovenous malformations: an update. Neurosurgery 34: Cunha e Sa MJ, Stein BM, Solomon RA, McCormick PC (1992) The treatment of associated intracranial aneurysms and arteriovenous malformations. J Neurosurg 77(6): Cushing, H., P. Bailey (1928) Tumours Arising from the Blood Vessels of the Brain. Angiomatous malformations and Hemangioblastomas, vol. III. Thomas. Springfield/Ill. p Dampsey RJ, Moftakhar R, Pozniak M (2004) Intraoperative Doppler to measure cerebrovascular resistance as a guide to complete resection of arteriovenous malformations. Neurosurgery 55(1): Dandy, W. E.: Venous abnormalities and angiomas of the brain (1928). Arch. Surg. (Chic.) 17: , 20. Deruty R, Pelissou-Guyotat I, Amat D, Mottolese C, Bascoulerque Y, Turiman F, Gerard JP (1996) Complications after multidisciplinary treatment of cerebral arteriovenous malformations. Acta Neurochir (Wien) 138(2): Dott, N. M (I933) Intracranial Aneurysms: Cerebral Arterio-Radiography: Surgical Treatments. Edinburgh Med. Jour 40, p Duong DH, Young WL, Vang MC, Sciacca RR, Mast H, Koennecke HC et al (1993) Feeding artery pressure and venous drainage pattern are primary determinants of hemorrhage from cerebral arteriovenous malformations. Stroke 29(6): Feliciano CE, de Leon-Berra R, Hernandez-Gaitan MS, Rodriguez-Mercado R (2010) A proposal for a new arteriovenous malformation grading scale for neuroendovascular procedures and literature review. P R Health Sci J 29(2): Fiorella D, Albuquerque FC, Woo HH, et al (2006) The role of neuroendovascular therapy for the treatment of brain arteriovenous malformations. Neurosurgery 59(5 suppl 3): Gao E, Young WL, Hademonos GJ, Massoud TF, Sciacca RR, Ma Q at al (1998) Theoretical modeling of arteriovenous rupture risk: a feasibility and validation study. Med Eng Phys 20(7): Green HD, Rapela CE (1964) Blood flow in passive vascular beds. Circ Res(Suppl 1);15: Guglielmi G (2008) Analysis of the hemodynamic characteristics of brain arteriovenous malformations using electric models: baseline settings, surgical extirpation, endovascular embolization and surgical bypass. Neurosurgery 63: Hacein-Bey L, Young WL (1999) Hemodynamic perturbations in cerebral arteriovenous malformations and management implications. Interv Neuroradiol 30(5): Hacein-Bey L, Young WL (1999) Hemodynamic perturbations in cerebral arteriovenous malformations and management implications. Interv Neuroradiol 30(5)(Suppl 1): Hademenos GJ, Massoud TF (1996) An electrical network model of intracranial arteriovenous malformations: analysis of variations in hemodynamic and biophysical parameters. Neurol Res 18(6): Hademonos GJ, Massoud TF, Vinuela F (1996) A biomathematical model of intracranial arteriovenous malformations based on electrical network analysis: theory and hemodynamics. Neurosurgery 38(5): Harvey, W (1953) De motu cordis (First english edition: G. Keynes. Nonsuch Press, London 33. Haw CS, terbrugge K, Willinsky R, Tomlinson G (2006) Complications of embolization of arteriovenous malformations of the brain. J Neurosurg 104(2): Hirai S, Mine S, Yamakami I, Ono J, Yamaura A (1998) Angioarchitecture related to hemorrhage in cerebral arteriovenous malformations. Neurol Med Chir (Tokyo).;38 Suppl: Hoffmann J (1898) Aneurysma racemosum der Gehirnarterien? Krankenvorstellung. Naturhistorisch-Medizinischer Verein, Heidelberg, 15. Juni Munch, med. Wschr. 2: 1159, 36. Hollerhage HG, Dewenter KM, Dietz H (1992) Grading of supratentorial arteriovenous malformations on the basis of multivariate analysis of prognostic factors. Acta Neurochir (Wien) 117(3 4): Hunter, W (1762) Further observations upon a particular species of aneurysm. Med. Observ. Enquir. (Lond.) 2: Irikura K, Morii S, Miyasaka Y, Yamada M, Tokiwa K, Yada K (1996) Impaired autoregulation in an experimental model of chronic cerebral hypoperfusion in rats. Stroke;27(8): Izawa M, Hayashi M, Chernov M, Nakaya K, Ochiai T, Murata N, Takasu Y, Kubo O, Hori T, Takakura K (2005) Long-term complications after gamma knife surgery for arteriovenous malformations. J Neurosurg;102 Suppl: Jafar JJ, Davis AJ, Berenstein A, Choi IS, Kupersmith MJ (1993) The effect of embolization with N-butyl cyanoacrylate prior to surgical resection of cerebral arteriovenous malformations. J Neurosurg 78: Jafar JJ, Rezai AR (1994) Acute surgical management of intracranial arteriovenous malformations. Neurosurgery 34:8 13, 42. Jahan R, Murayama Y, Gobin YP, et al (2001) Embolization of arteriovenous malformations with Onyx: clinicopathological experience in 23 patients. Neurosurgery;48: Karlsson B, Lindquist C, Steiner L (1996) Effect of gamma knife surgery on the risk of rupture prior to AVM obliteration. Minim Invasive Neurosurg 39: Kerber CW, Hecht ST, Knox K (1997) Arteriovenous malformation model for training and research. Am J Neuroradiol 18(7): Krings T, Hans FJ, Geibprasert S, Terbrugge K (2010) Partial targeted embolisation of brain arteriovenous malformations. Eur Radiol. Jun 11. DOI: /s Lawton MT, Kim H, McCulloch CE, Mikhak B, Young WL (2010) A supplementary grading scale for selecting patients with brain arteriovenous malformations for surgery. Neurosurgery 66(4): Ledezma CJ, Hoh BL, Carter BS, Pryor JC, Putman CM, Ogilvy CS (2006) Complications of cerebral arteriovenous embolization: multivariate analysis of predictive factors. Neurosurgery 58(4): Leksell L (1968) Cerebral radiosurgery. I. Gammathalanotomy in two cases of intractable pain. Acta Chir Scand 134(8): Lindquist C (1996) Comment on Hemorrhage risk after stereotactic radiosurgery of cerebral arteriovenous malformations. Neurosurgery 38: Lindqvist M, Karlsson B, Guo WY, Kihlstrom L, Lippitz B, Yamamoto M (2000) Angiographic long-term follow-up data for arteriovenous malformations previously proven to be obliterated after gamma knife radiosurgery. Neurosurgery 46: Lo EH (1993) A theoretical analysis of hemodynamic and biomechanical alteration in intracranial arteriovenous malformations after radiosurgery. Int J Radiat Oncol Biol Phys 27(2): Lo EH, Fabrikant JI (1992) A compartmental flow model for intracranial AVMs. In: Lunsford LD, ed. Stereotactic Radiosurgery Update. New York, NY: Elsevier Lo EH, Fabrikant JI, Levy RP, Phillips MH, Frankel KA, Alpen EL (1991) An experimental compartmental flow model for assessing the hemodynamic response of intracranial arteriovenous malformations to stereotactic radiosurgery. Neurosurgery 28: Luessenhop AJ, Spence WT (1960) Artificial embolization of cerebral arteries. Report of use in a case of arteriovenous malformation. J Am Med Assoc. Mar 12;172: Luschka, H (1854) Cavernose Blutgeschwulst des Gehirns. Virchows Arch. 6: Malpighi, M (1686) De pulmonibus in opuscula anatomica, Bologna, 1661.Opera omnia. Scott, London (vol. I and II) 24 25
14 57. Marks MP, Lane B, Steinberg G, Chang P (1991) Vascular characteristics of intracerebral arteriovenous malformations in patients with clinical steal. Am J Neuroradiol 12(3): Maruyama K, Kawahara N, Shin M, Tago M, Kishimoto J, Kurita H, et al (2005) The risk of hemorrhage after radiosurgery for cerebral arteriovenous malformations. N Eng J Med 352: Massoud TF, Hademonos GJ, De Salles AA, Kolberg TD (2000) Experimental radiosurgery simulations using a theoretical model of cerebral arteriovenous malformations. Stroke 31(10): Mast H, Mohr JP, Osipov A, Pile-Spellman J, Marshall RS, Lazar RM et al (1995). Steal is an unestablished mechanism for the clinical presentation of cerebral arteriovenous malformations. Stroke 26(7): Matsumoto H, Takeda T, Kohno K, Yamaguchi Y, Kohno K, Takechi A et al (2006) Delayed hemorrhage from completely obliterated arteriovenous malformation after gamma knife radiosurgery. Neurol Med Chir (Tokyo) 46(4): McCormick WF. The pathology of vascular ( arteriovenous ) malformations (1966) J Neurosurg;24: Miyasaka Y, Yada K, Owada T, Kitahara T, Kurata A, Irikura K (1992) An analysis of the venous drainage system as a factor in hemorrhage from arteriovenous malformations. J Neurosurg 76(2): Moniz, E (1927) L encephalographie arterielle, son importance dans la localization des tumeurs cerebrales. Rev. neurol. 2: 72-89, 65. Moreno-Jimenez S, Celis MA, Larraqa-Gutierrez JM, Suarez-Campos Jde J, Garcia-Gardunno A, Hernandez-Bojorquez M, Gutierrez-Aceves GA (2007) Intracranial arteriovenous malformations treated with LINAC-based conformal radiosurgery: validation of the radiosurgery-based arteriovenous malformation score as a predictor of outcome. Neurol Res 29(7): Morgan M, Winder M (2001) Haemodynamics of arteriovenous malformations of the brain and consequences of resection. J Clin Neurosci 8(3): Muacevic A, Steiger HJ (1999) Computer-assisted resection of cerebral arteriovenous malformations. Neurosurgery 45(5): Murphy JP (1954) Cerebrovascular Disease, vol. V. Yearbook Medical Publishers, Chicago pp Nataf F, Meder JF, Roux FX, Blustajn J, Merienne L, Merland JJ, Schlienger M, Chodkiewicz JP (1997) Angioarchitecture associated with haemorrhage in cerebral arteriovenous malformations: a prognostic statistical model. Neuroradiology 39(1): Nicholas WW, O Rourke MF (1990) McDonald s Blood Flow in Arteries. Theoretic, Experimental and Clinical Principles, third edn, London Edward Arnold,: Nornes H, Grip A (1980) Hemodynamic aspects of cerebral arteriovenous malformations. J Neurosurg 53: Ogilvy CS (1990) Radiation therapy for arteriovenous malformations: a review. Neurosurgery 26: Ogilvy CS, Stieg PE, Awad I, Brown Jr RD, Kondziolka D, Rosenwasser R, Young WL, Hademonos G (2001) Recommendations for the Management of Intracranial Arteriovenous Malformations: A statement for Healthcare Professionals From a Special Writing Group of the Stroke Council American Stroke Association. Stroke 32: Perata HJ, Tomsick TA, Tew JM (1994) Feeding artery pedicle aneurysms: association with parenchyma hemorrhage and arteriovenous malformation In the brain. J Neurosurg 80: Pérez-Higueras A, Rossi Lopez R, Quinones Taria D (2005) Endovascular treatment of cerebral AVM: our experience with Onyx. Interventional Neuroradiology 11: Pertuiset B, Ancri D, Kinuta Y, et al (1991) Classification of supratentorial arteriovenous malformations. A score system for evaluation of operability and surgical strategy based on an analysis of 66 cases. Acta Neurochir (Wien). 110(1 2): Picard L, Da Costa E, Anxionnat R, Macho J, Bracard S, Per A, Marchal JC (2001) Acute spontaneous hemorrhage after embolization of brain arteriovenous malformation with N-butyl cyanoacrylate. J Neuroradiol 28: , 78. Pollock BE (2000) Occlusive hyperemia: a radiosurgical phenomenon? Neurosurgery 47(5): Pollock BE, Flickinger JC (2002) A proposed radiosurgery-based grading system for arteriovenous malformations. J Neurosurg 96(1): Pollock BE, Flickinger JC, Lunsford LD, Bissonette DJ, Kondziolka D (1996) Hemorrhage risk after stereotactic radiosurgery of cerebral arteriovenous malformations. Neurosurgery 38: Raffa SJ, Chi YY, Boya FJ, Friedman WA (2009) Validation of the radiosurgery-based arteriovenous malformation score in a large linear accelerator radiosurgery experience. J Neurosurg 111(4): Redekop G, TerBrugge K, Montanera W, Willinsky R (1998) Arterial aneurysms associated with cerebral AVMs: classification, incidence, and risk of hemorrhage. J Neurosurgery 89(4): Rokitansky, C (1846) Handbuch der allgemeinen pathologischen Anatomie. Wien p Sanchez-Mejia RO, McDermott MW, Tan J, Kim H, Young WL, Lawton MT (2009) Radiosurgery facilitates resection of brain arteriovenous malformations and reduces surgical morbidity. Neurosurgery 64(2): Sekhon LH, Morgan MK, Spence I, Weber NC (1994) Chronic cerebral hypoperfusion and impaired neuronal function in rats. Stroke 25(5): Sheth RD, Bodensteiner JB (1995) Progressive neurologic impairment from an arteriovenous malformation vascular steal. Pediatr Neurol 13(4): Shin M, Kawamoto S, Kurita H, Tago M, Sasaki T, Morita A, Ueki K, Kirino T (2002). Retrospective analysis of a 10-year experience of stereotactic radiosur- gery for arteriovenous malformations in children and adolescents. J Neurosurg 97(4): Sorimachi T, Takeuchi S, Koike T, Minakawa T, Abe H, Tanaka R (1995) Blood pressure monitoring in feeding arteries of cerebral arteriovenous malformations during embolization: a preventive role in hemodynamic complications. Neurosurgery 37(6): Spears J, Terbrugge KG, Moosavian M, et al (2006) A discriminative prediction model of neurological outcome for patients undergoing surgery of brain arteriovenous malformations. Stroke 37(6): Spetzler RF, Martin NA (1986) A proposed grading system for arteriovenous malformations. J Neurosurg 65(4): Spetzler RF, Martin NF, Carter LP, Flom RA, Raudzens PA, Wilkinson E (1987) Surgical management of large AVMs by staged embolization and operative excision. J Neurosurg 67: Steiner L, Lindquist C, Adler JR, Torner JC, Alves W, Steiner M (1992) Clinical outcome of radiosurgery for cerebral arteriovenous malformations. J Neurosurg 77(1): Tamaki N, Ehara K, Lin TK, et al. Cerebral arteriovenous malformations: factors influencing the surgical difficulty and outcome. Neurosurgery 1991;29(6): Taylor CL, Dutton K, Rappard G, Pride GL, Replogle R, Purdy PD, White J, Giller C, Kopitnik TA, Samson DS (2004) Complications of preoperative embolization of cerebral arteriovenous malformations. J Neurosurg 100: , 95. Tevah J, Huete I (2005) Endovascular treatment of cerebral AVMs with a new material: Onyx. Interventional Neuroradiology 11: Thompson RC, Steinberg GK, Levy RP, Marks MP (1998) The management of patients with arteriovenous malformations and associated intracranial aneurysms. Neurosurgery 43(2): Todaka T, Hamada J, Kai Y, Morioka H, Ushio Y (2003) Analysis of mean transit time of contrast medium in ruptured and unruptured arteriovenous malformations: a digital subtraction angiographic study. Stroke 34(10): Tu J, Karunanayaka A, Windsor A, Stoodley MA (2010) Comparison of an animal model of arteriovenous malformation with human arteriovenous malformation. J Clin Neurosci 17(1): Ujiie H, Tamano Y, Xiuling L, Hori T (2000) Haemorrhagic complications after total extirpation of huge arteriovenous malformation. J Clin Neurosci.; 7 Suppl 1: Valavanis A, Christoforidis G (1999) Endovascular management of cerebral arteriovenous malformations. Neurointerventionist;1: Viñuela F, Dion JE, Duckwiler G, Martin NA, Lylyk P, Fox A et al (1991) Combined endovascular embolization and surgery in the management of cerebral arteriovenous malformations: Experience with 101 cases. J Neurosurg 75: , 102. Viñuela F, Duckwiler G, Guglielmi G (1997) Contribution of interventional neuroradiology in the therapeutic management of brain arteriovenous malformations. J Stroke Cerebrovasc Dis 4: Vinuela F, Nombela L, Roach MR, Fox AJ, Pelz DM (1985) Stenotic and occlusive disease of the venous drainage system of deep brain AVMs. J Neurosurg 63(2): Virchow. R (1854) Uber cavernbse (erectile) Geschwiilste und Teleangiektasien.Virchows Arch. path. Anat. 6: Wakhloo AK, Lieber BB, Siekmann R, Eber DJ, Gounis MJ (2005) Acute and chronic swine rete arteriovenous malformation models: hemodynamice and vascular remodeling. Am J Neuroradiol 26(7): Wallace RC, Flom RA, Khayata MH, Dean BL, McKenzie J, Rand JC et al (1995) The safety and effectiveness of brain arteriovenous malformation embolization using acrylic and particles: the experience of a single institution. Neurosurgery 37(4): Yasargil MG (1987) Microneurosurgery IIIA. Thieme New York, p Young WL, Kader A, Pile-Spellman J, Ornstein E, Stein BM (1994) Arteriovenous malformation draining vein physiology and determinants of transnidal pressure gradients. The Columbia University AVM Study Project. Neurosurgery.;35(3): Young WL, Pile-Spellman J, Prohornik I, Kader A, Stein BM (1994) Evidence for adaptive autoregulatory displacement in hypotensive cortical territories adjacent to arteriovenous malformations. Columbia University AVM Study Project. Neurosurgery 34(4): Yuki I, Kim RH, Duckwiler G, Johan R, Tateshima S, Gonzales N, et al (2009) Treatment of brain arteriovenous malformations with high-flow arteriovenous fistulas: risk and complications associated with endovascular embolization in multimodality treatment. J Neurosurg.;16 DOI: / JNS
15 MANAGEMENT BASIC DIAGNOSTIC METHODS IN DERMATOLOGY PART I DERMATOSCOPY AND VIDEODERMATOSCOPY ZUZANNA ŁAGUN, MARTA WIECZOREK, ELŻBIETA SZYMAŃSKA, IRENA WALECKA The Dermatology Department of the Central Clinical Hospital of the Ministry of the Interior, Warsaw, Poland Over the past few years there has been a great increase in the number of skin cancers. This mainly refers to melanoma, as well as basal cell carcinoma and squamous cell carcinoma, which is accompanied by enormous costs in medical care and a significant mortality rate, especially in relation to melanoma. Fortunately, there are simple and effective diagnostic methods enabling early detection of skin cancer by visual examination of skin lesions. It is essential for doctors of other specialisations, mainly for general practitioners to refer to diagnostic procedures used by dermatologists, which have a great impact on the patient s life and health [2]. Through the use of dermatoscopy, an experienced doctor can contribute to more effectiveclinical evaluation of melanotic, non-melanotic, benign and malignant skin lesions, resulting in much better recognizability of melanoma versus clinical evaluation itself. Furthermore, dermatoscopy simplifies diagnosis of extravasations, foreign bodies and even human scrabies, as well as being very effective in the diagnosis of subungual abnormalities [2, 3]. Abstract The skin is one of the largest and most important human organs. It covers and protects the organism, performing numerous functions including those protective, representative and social. In most cases, skin diseases are not life-threatening, although they often cause pain and suffering, discomfort, disfigurement, embarrassment and social stigmatism. In primary care, up to 25 % of visits involve skin disorders. Therefore, it is crucial for general practitioners to have the basic knowledge of skin disorders, as well as support of the right equipment to enable them to make the right diagnosis, choose the best treatment methods or recognize those needing referral to specialist services due to diagnostic difficulties. This mainly applies to skin lesions, which are benign in most cases, however skin cancer is associated with significant morbidity and mortality and therefore visual examination of the entire cutaneous surface may have a positive impact on the outcomes for the patient. Techniques such as dermatoscopy and videodermatoscopy may offer positive assistance. With appropriate training and regular use of these methods by dermatologists, as well as other specialists, dermatoscopy and videodermatoscopy may prove very useful [1, 6]. Dermatoscopy The dermatoscope is a device that illuminates the skin and by using optical properties allows the doctor to visualise the color and the structure of the epidermis, derma- -epidermal junction, as well as the papillomatous layer of epidermis - things we cannot visualize with the naked eye. Classic dermatoscopy examination of skin lesions requires prior application of immersion oil, liquid or gel and a close link to the glass surface of the dermatoscope. The liquid eliminates reflection of the light and, as a result, the horn layer of the epidermis becomes pellucid, enabling the appearance of the colors and structures localized under the epidermis. During dermatoscopic analysis, it is necesary to observe the following rules. First of all, it has to be decided whether the skin lesion is pigmented or non-pigmented. Thereafter, by using the ABCD rule (A-assymetry, B-border, C-color, D- diameter or differential structures) the skin lesion may be evaluated, assigned relevant points and qualified as benign, suspicious or malignant. Consecutively, the TDS (Total Dermatoscopy Score) is assigned. The greater the assymetry, the more irregular the border, the greater the colour range and number of structures seen under the dermatoscope, the higher the TDS score [2, 3]. Dermatoscopy is a method not only intended for use by dermatologists. Every doctor, despite theirprofession may use dermatoscopy to assess whether a suspicious skin lesion should be looked at by a more experienced clinicist. When one starts to use the dermatoscope,it is essential to decide, whether the skin lesion looks suspicious and the patient should consult witha dermatologist, who will decide on eitheraskin biopsy or surgical removal of the lesion. It is important to remember that dermatoscopy is not a replacement for histopathological examination [6]. Videodermatoscopy One of the most innovative forms of dermatoscopy is videodermatoscopy.it is a diagnostic method for pigmented skin lesions, combining an optical system using a videocamera to digitally analyse the image on the monitor. To evaluate skin moles, zooming of 20-70x may be used and the time needed to examine the patient is approximately minutes. Non-invasive examination and the possibility of reviewing images at any time are significant advantages of this method. During examination, the skin lesion is covered with a liquid substance which better enables the viewing of deeper layers of the epidermis. Thereafter, the picture is electronically saved with the videocamera. Every diagnosed naevus stays in RAM and as a result it can be easily ascertained during reexamination whether the pigmented naevus has progressed. Videodermatoscopy enables detection of new or changed naevi including their evaluation, long-term observation, the possibility of creating a data base, comparing the naevi during subsequent monitoring together with their treatement, whereby it is easier to observe such lesions rather than depend upon the evaluator s memory. The device itself, along with computer programs, does not enable a diagnosis to be made. An accurate diagnosis alwaysdepends upon the assessment of an experienced doctor [4, 5]. Videodermatoscopes also enable a nailfold capillaroscopy to be performed. This may be useful in diagnosing systemic connective tissue diseases as well as performing a precise analysis of the scalp. Trichoscopy (in other words dermatoscopic analysis of the scalp) is a very meaningful examination, which assists other diagnostic methods (trichogram and histopathological examination) and is used in determining the causes of alopecia-scaring and non-scaring, as well as in some congenital disorders of the hair shaft structures [4]. Summary Basic training in using a dermatoscope and videodermatoscope requires only a few days, however like other diagnostic methods, in order to achieve an adequate level of proficiency, constant practice and application of the method on a regular basis are essential. Without appropriate training, the diagnostic accuracy of dermatoscopy matches the estimate of the naked eye. Dermatoscopy is very useful to those, who know how to make use of this procedure. [6] n 28 29
16 Bibiography 1. Dermatological history and examination Shalini Narayan Dermatological history and examination, Z, Volume 41, Issue 6, Pages , Copyright Dermoscopy Ashfaq A. Marghoob, Azadeh Esmaeili and Alon Scope Pfenninger and Fowler s Procedures for Primary Care, Chapter 15, Dermatoscopy Wilhelm Stolz Braun-Falco Dermatology, Chapter 5, Dermoscopy Ignazio Stanganelli, MD; Chief Editor: Dirk M Elston, MD 5. Tips and tricks in the dermoscopy of pigmented lesions Grażyna Kamińska-Winciorek and Radoslaw Spiewak 6. Dermoscopy for the Family Physician Ashfaq A. Marghoob, MD, Memorial Sloan-Kettering Cancer Center, New York, New York Richard P. Usatine, MD, University of Texas Health Science Center, San Antonio, Texas Natalia Jaimes, MD, Aurora Skin Cancer Center and Universidad Pontificia Bolivariana, Medellín, Colombia Am Fam Physician Oct 1;88(7): ROLA I MIEJSCE CHEMIOTERAPII W LECZENIU SKOJARZONYM NOWOTWORÓW GÓRNEGO ODCINKA UKŁADU POKARMOWEGO ANDRZEJ DEPTAŁA Klinika Onkologii i Hematologii, Centralny Szpital Kliniczny MSW, Warszawa, Polska. Kierownik Kliniki: Prof. dr hab. med. Andrzej Deptała Zakład Profilaktyki Onkologicznej, Warszawski Uniwersytet Medyczny, Warszawa, Polska. Adres do korespondencji: Andrzej Deptała Klinika Onkologii i Hematologii, Centralny Szpital Kliniczny MSW, ul. Wołoska 137, Warszawa, Polska, tel./fax: / , andrzej.deptala@cskmswia.pl Nowotwory górnego odcinka układu pokarmowego, a więc guzy wywodzące się pierwotnie z przełyku, żołądka, jelita cienkiego, trzustki, wątroby i dróg żółciowych, to różnorodna grupa chorób pod względem biologii, przebiegu klinicznego, rokowania oraz wyników leczenia. Chemioterapia (klasyczna oraz celowana molekularnie) odgrywa istotną rolę w skojarzonym leczeniu tego typu nowotworów, zarówno postaci ograniczonych, jak również zaawansowanych miejscowo, a w przerzutowych stanowi podstawę postępowania. Praca na temat roli i miejsca chemioterapii w skojarzonym leczeniu nowotworów (tj. raków, chłoniaków, mięsaków, guzów neuroendokrynnych, czerniaka, itd.) górnego odcinka układu pokarmowego zajęła by swą objętością pokaźną książkę. Autor pisząc ten artykuł, był zmuszony do ograniczenia omówienia tego zagadnienia w leczeniu skojarzonym najczęstszych nowotworów i w tych sytuacjach klinicznych, w których w ostatnim okresie i przy współudziale chemioterapii uzyskano największe korzyści w postaci poprawy przeżycia. Dlatego, w dalszej części pracy zostały przestawione aktualne poglądy na temat postępowania w operacyjnych rakach przełyku, połączenia przełykowożołądkowego i żołądka przy zastosowaniu chemioterapii (CTH) oraz chemioradioterapii (CTH-RTH). Streszczenie: Nowotwory górnego odcinka układu pokarmowego to różnorodna grupa chorób pod względem biologii, przebiegu klinicznego, rokowania oraz wyników leczenia. Chemioterapia (klasyczna oraz celowana molekularnie) odgrywa istotną rolę w skojarzonym leczeniu tego typu nowotworów, zarówno postaci ograniczonych, jak również zaawansowanych miejscowo. Praca na temat roli i miejsca chemioterapii w skojarzonym leczeniu nowotworów (tj. raków, chłoniaków, mięsaków, guzów neuroendokrynnych, czerniaka, itd.) górnego odcinka układu pokarmowego (GOUP) zajęła by swą objętością pokaźną książkę. Autor tego artykułu ograniczył się więc do omówienia najczęstszych postaci i w sytuacjach klinicznych, w których w ostatnim okresie uzyskano największe korzyści mierzone poprawą przeżyć tj. w operacyjnych rakach: przełyku, połączenia przełykowo-żołądkowego i żołądka. Słowa kluczowe: skojarzone leczenie, chemioterapia okołooperacyjna, chemioradioterapia, rak przełyku, rak połączenia przełykowo-żołądkowego, rak żołądka Rak przełyku i rak połączenia przełykowo-żołądkowego W Polsce w 2012 r. stwierdzono 1256 zgonów z powodu raka przełyku u mężczyzn i 319 u kobiet [1]. Podstawą wspólnego omówienia raka przełyku (EC esophageal cancer) i raka połączenia przełykowo-żołądkowego (EJC esophagogastric-junction cancer) stało się włączenie EJC do systemu AJCC/UICC (American Joint Committee on Cancer/Unio Internationale Contra Cancrum) oceny zaawansowania klinicznego raka przełyku oraz leczenie chorych na EC i EJC tymi samymi protokółami terapeutycznymi w ramach prospektywnych, randomizowanych badań klinicznych, z losowym doborem chorych. Wyniki leczenia chirurgicznego raka przełyku, mimo postępu w chirurgii, są w dalszym ciągu niezadowalające. Po ezofagektomii 5-letnie przeżycie w guzach w II i III stopniu zaawansowania wg AJCC, uzyskuje odpowiednio, około 45% i 20% pacjentów z rakiem płaskonabłonkowym (SCC squamous cell carcinoma) oraz około 40% i 19% pacjentów z gruczolakorakiem (ADC adenocarcinoma) [2]. Stąd też poprawę przeżycia chorych upatruje się w zastosowaniu okołooperacyjnej chemioterapii lub chemioradioterapii. Przełomowe, w leczeniu skojarzonym EC i EJC, wydawały się być wyniki randomizowanego i prospektywnego badania III fazy z losowym doborem chorych CROSS II [3]. W badaniu tym poddano ostatecznej analizie 366 chorych na EC (76%) i EJC (24%), z których 75% (275) miało rozpoznanie ADC, 23% (84) SCC, a 2% (7) raka niezróżnicowanego wielkokomórkowego. Pacjentów z kliniczne ustalonym zaawansowaniem choroby, jako T1N1 i T2-3/ N0-1 (IIA, IIB i IIIA wg AJCC) przydzielono losowo do grupy z przedoperacyjną (neoadiuwantową) chemioradioterapią (paklitaksel/karboplatyna + 41,4 Gy w 23 frakcjach) z następową resekcją (ramię CTH-RTH-CH) lub do grupy leczonej wyłącznie chirurgicznie (ramię CH). Analiza zgodna z zaplanowanym leczeniem (ITT intention-to-treat) wykazała, że w ramieniu CTH-RTH-CH mediana przeżycia całkowitego (OS overall survival) była dwukrotnie dłuższa niż w ramieniu CH i wyniosła 49,4 miesięcy w porównaniu z medianą 24 miesięcy u chorych leczonych wyłącznie chirurgicznie (p = 0,003; ryzyko hazardu [HR hazard risk], 0,657; 95% CI, 0,495-0,871). Przeżycie dwu- i 5-letnie w każdym z ramion 30 31
17 MANAGEMENT uzyskało odpowiednio 67% i 47% oraz 50% i 34% chorych. Większą poprawę OS po zastosowaniu przedoperacyjnej radiochemioterapii uzyskali chorzy z podtypem SCC (p = 0,011; skorygowany HR, 0,422; 95% CI, 0,226-0,788; skorygowany względem HR p = 0,007) niż pacjenci, u których rozpoznano ADC (p = 0,049; skorygowany HR, 0,741; 95% CI, 0,536-1,024; skorygowany względem HR p = 0,07). Resekcję chirurgiczną przeprowadzono u 94% chorych z ramienia CTH-RTH-CH i u 99% z ramienia CH (p = 0,01), jednakże wycięcie typu R0 udało się wykonać odpowiednio, u 92% vs 69% (p < 0,0001) chorych, a całkowitą odpowiedź patologiczną (ypt0n0) uzyskano u 29% i jedynie w ramieniu CTH-RTH-CH (u 23% z ADC i u 49% z SCC; p = 0,008). Leczenie było dobrze tolerowane, a poważne objawy niepożądane związane z chemioradioterapią wystąpiły u kilku procent pacjentów (najczęstsze: leukopenia u 6%, małopłytkowość u 2%, jadłowstręt u 5%, męczliwość u 3%). Śmiertelność w okresie pooperacyjnym w czasie pobytu w szpitalu dotyczyła 4% chorych w każdym z ramion, a 30-dniowa 2% w ramieniu CTH-RTH-CH i 3% przy samym leczeniu chirurgicznym. Nieszczelność zespolenia wystąpiła odpowiednio, u 22% i 30% chorych [3]. W 2014 r. ten sam zespół badaczy holenderskich dokonał analizy badań CROSS I i CROSS II (odpowiednio, II i III fazy) pod kątem typów nawrotu operacyjnego EC/EJC w ramionach CTH-RTH-CH vs CH [4]. Analizie poddano 418 chorych (75% z rozpoznaniem gruczolakoraka), która wykazała, że zastosowanie przedoperacyjnej CTH-RTH zmniejszyło ogólną częstość nawrotu raka do 34% (z 58% w ramieniu CH) i odpowiednio częstość: wznów miejscowych (lokoregionalnych) do 14% z 34% (p < 0,001), rozsiewu otrzewnowego do 4% z 14% (p < 0,001), rozsiewu krwiopochodnego do 29% z 34% (p < 0,025) [3]. Jednakże po publikacji powyżej analizy, w listach do redakcji Journal of Clinical Oncology, podniosły się głosy krytyczne, bowiem inni badacze zauważyli błędy interpretacyjne, których dopuścili się autorzy badań CROSS. Najpoważniejsze wątpliwości wysunęli Smyth i wsp. w liście do edytora, tytułując przewrotnie: Should we be CROSS? [5]. Ich zastrzeżenia do badań CROSS były następujące: 1/nie uzyskano statystycznie znamiennego wydłużenia OS pod wpływem CTH-RTH u chorych z rozpoznaniem operacyjnego gruczolakoraka (75% chorych w badaniu CROSS; p = 0,07); 2/nie uzyskano statystycznie znamiennego wydłużenia OS pod wpływem CTH-RTH u chorych z klinicznymi cechami zajęcia węzłów chłonnych przed rozpoczęciem leczenia (65% chorych w badaniu CROSS; skorygowany HR, 0,806; 95% CI, 0,576-1,130; skorygowany względem HR p = 0,21); 3/chemioradioterapia nie spowodowała zmniejszenia ryzyka wystąpienia izolowanych przerzutów odległych; 4/nie przedstawiono analizy przerzutów odległych w korelacji z histologicznym typem nowotworu; 5/błędnie zaklasyfikowano przerzuty do węzłów chłonnych nadobojczykowych, jako nawroty lokoregionalne, podczas gdy wg klasyfikacji AJCC [2] są to przerzuty odległe (cecha M1). Dlatego Smyth i wsp. skonkludowali, że jedyną grupą, która odniosła rzeczywiste korzyści w przeżyciu Tabela I. PIĘCIOLETNIE PRZEŻYCIA PODGRUP CHORYCH W BADANIU ACTS-GC [WG 11] OS (%) RFS (%) S-1 CH S-1 CH II 84,2 71,3 79,2 64,4 IIIA 67,1 57,3 61,4 50,0 IIIB 50,2 44,1 37,6 34,4 Ryzyko hazardu (HR) dla: OS RFS 0,509; 95% CI, 0,338-0,765 0,521; 95% CI, 0,362-0,750 0,708; 95% CI, 0,510-0,983 0,696; 95% CI, 0,514-0,941 0,791; 95% CI, 0,520-1,205 0,788; 95% CI, 0,539-1,151 z neoadiuwantowej CTH-RTH byli chorzy na operacyjnego raka płaskonabłonkowego z kliniczną cechą N0 [5]. W 2014 r. autorzy brytyjscy [6] przeprowadzili analizę (na podstawie prospektywnie zebranych danych z lat ) 584 resekcji z powodu gruczolakoraka EC i EJC, w której udowodnili, że zmniejszenie stopnia zaawansowania nowotworu (T/N downstaging) za pomocą chemioterapii neoadiuwantowej (ECF epirubicyna, cis platyna, 5-FU) z następową operacją statystycznie znamiennie poprawia przeżycie chorych (p < 0,001) w porównaniu z osobami, u których nie uzyskano odpowiedzi na CTH przedoperacyjną i to zmniejszenie zaawansowania okazało się najsilniejszym niezależnym dodatnim czynnikiem predykcyjnym dla OS (HR, 0,43; 95% CI, 0,31-0,59). Ponadto wykazano, że u chorych odpowiadających redukcją zaawansowania nowotworu na przedoperacyjną chemioterapię, w porównaniu do nieodpowiadających, stwierdza się zmniejszenie ryzyka nawrotu lokoregionalnego (odpowiednio, 6% vs 13%; p = 0,030) i wznowy w postaci pojawienia się przerzutów odległych (odpowiednio, 9% vs 29%; p = 0,027) [6]. Podsumowując tę część artykułu, należy stwierdzić, że chorzy z operacyjnym płaskonabłonkowym rakiem przełyku w II i III stopniu klinicznego zaawansowania wg AJCC powinni być leczeni za pomocą chemioradioterapii przedoperacyjnej z użyciem paklitakselu i karboplatyny, z następową radykalną resekcją chirurgiczną. W przypadku pacjentów z operacyjnym gruczolakorakiem przełyku i połączenia przełykowo-żołądkowego, o tym samym klinicznym zaawansowaniu choroby, nie można jednoznacznie uznać podobnej strategii za optymalną, bowiem zarówno badania CROSS, jak i wcześniejsze badanie Stahla i wsp. [7] (różnice w OS wypadły w nim niezamiennie statystycznie) nie dostarczyły przekonujących dowodów na jej potwierdzenie. Niewątpliwie równorzędną alternatywą dla chemioradioterapii przedoperacyjnej, u pacjentów z ADC EC/EJC w II i III stopniu zaawansowania wg AJCC, jest zastosowanie okołooperacyjnej chemioterapii (jak w raku żołądka). Tabela II. PORÓWNANIE WYNIKÓW 3-LETNICH PRZEŻYĆ W BADANIACH ACTS-GC, INT0116 I MAGIC [ZMODYFIKOWANO WG 8] ACTS-GC INT0116 MAGIC Chorzy leczeni wyłącznie chirurgicznie 3-letnie OS 3-letni RFS/PFS Chorzy leczeni z dodaniem chemio- (radio)terapii 3-letnie OS 3-letni RFS/PFS Ryzyko hazardu między ramionami Zgonu Progresji 70,1 59,6 80,1 72,2 0,68 0,62 41,0 31,0 50,0 48,0 0,74 0,66 Rak żołądka Rak żołądka (GC gastric cancer) jest czwartym co do częstości zachorowania nowotworem na świecie, a drugim pod względem liczby zgonów [2]. W Polsce, w 2012 r., w strukturze zgonów zajął 4 miejsce u mężczyzn (6,6%) i 6 u kobiet (4,4%), a standaryzowane współczynniki umieralności wyniosły odpowiednio, 11,3/105 i 4,1/105. W Polsce mimo stałego spadku (od lat 70-tych) liczby zachorowań i zgonów z powodu raka żołądka, w 2012 r. zmarło z powodu tego nowotworu aż 5317 osób (3475 mężczyzn i 1842 kobiety) [1], co oznacza, że rak ten był zbyt późno rozpoznany i większość chorych było kandydatami do leczenia skojarzonego lub paliatywnego. Leczenie wyłącznie chirurgiczne jest zarezerwowane dla wczesnych postaci GC, natomiast chorzy z operacyjnym rakiem miejscowo zaawansowanym (stadium II i wyższe wg AJCC, ale koniecznie z cechą M0 [2]) mogą skorzystać z jednej z 3 opcji leczenia skojarzonego: chemioterapia okołooperacyjna (CTHO), chemioterapia adiuwantowa (CHTA) i chemioradioterapia adiuwantowa (CTH-RTH). Pierwsza opcja dominuje w Zachodniej Europie, druga w Japonii, Korei i Chinach, a trzecia w Stanach Zjednoczonych Ameryki [8]. Merytoryczne uzasadnienie do zastosowania CTHO oparte jest na wynikach dwóch badań prospektywnych i randomizowanych III fazy, z losowym doborem chorych brytyjskiego MAGIC (w badaniu znaleźli się też chorzy z Brazylii, Holandii, Niemiec, Nowej Zelandii i Singapuru) [9] i francuskiego ACCORD-07 [10]. Do badania MAGIC Cunningham i wsp. [9] zakwalifikowali i poddali analizie ITT 503 chorych na operacyjnego gruczolakoraka żołądka (74%), gruczolakoraka połączenia przełykowo-żołądkowego (około 12%) i gruczolakoraka dolnego odcinka przełyku (około 15%), których losowo przydzielono do ramienia z CTHO (250) i do ramienia leczonego wyłącznie chirurgicznie (CH 253 osoby). Chemioterapia składała się z epirubicyny, cis platyny i 5-FU (schemat ECF), podawanych co 21 dni. Trzy cykle CTHO podano przed zabiegiem operacyjnym, a kolejne 3 po operacji, którą wykonano u 91,6% chorych. W ramieniu CH resekcję wykonano u 96,4% pacjentów. Zakres operacji zależał od chirurga i dlatego limfadenektomię D1 wykonano aż u około 20% chorych, a limfadenektomię D2 u około 40% (nie podano zakresu limfadenektomii w EC). W badaniu histopatologicznym po operacji, w ramieniu CH znaleziono znacznie więcej guzów pt3/t4 (65%) i większą liczbę przerzutowych węzłów chłonnych (pn2/n3 30%) w porównaniu do ramienia z CTHO (odpowiednio, 48% i 16%), co zostało przypisane korzystnemu wpływowi chemioterapii na zmniejszenie wielkości nowotworu (downstaging). W ramieniu CTHO pełne leczenie ukończyło jedynie 42% chorych (34% pacjentów 31,0 25,0 44,0 40,0 0,75 0,66 nie otrzymało po operacji chemioterapii z powodu progresji, rezygnacji chorego lub powikłań chirurgicznych). Przy medianie obserwacji wynoszącej 49 miesięcy wykazano statystycznie istotne zmniejszenie prawdopodobieństwa progresji choroby (PFS progression free survival) pod wpływem CTHO (HR, 0,66; 95% CI, 0,53-0,81; p < 0,001) oraz ryzyka zgonu (HR, 0,75; 95% CI, 0,60-0,93; p = 0,009). Przeżycie 5-letnie (OS) wzrosło do 36,3% w ramieniu CTHO w stosunku do 23% w ramieniu CH. Poważne objawy niepożądane (w stopniu 3 i 4) związane z chemioterapią przed- i pooperacyjną wystąpiły u 11% chorych, pod postacią neutropenii (24 i 28%), limfocytopenii (17 i 20%), nudności (6 i 12%), wymiotów (6 i 10%), zapalenia jamy ustnej (4%) i biegunki (3%) [9]. Badanie francuskie ACCORD-07 [10] różniło się od badania MAGIC mniejszą ogólną liczbą chorych (224 vs 503), schematem chemioterapii (cis platyna i 5-FU, podawane co 28 dni vs ECF co 21 dni) i znacznie większą liczbą chorych na EJC (144 65% vs 58 12%), a mniejszą z GC (55 25% vs %). Poza powyższymi, założenia, cele, struktura i punkty końcowe obu badań były takie same. Uzyskane wyniki wypadły niemal identycznie. Zabieg chirurgiczny wykonano u 96,5% chorych otrzymujących chemioterapię i u 99% w grupie leczonej wyłącznie operacyjnie. W ramieniu CTHO, w porównaniu z CH, 5-letnie przeżycie całkowite uzyskało istotnie więcej chorych 38% vs 24% (HR dla zgonu, 0,69; 95% CI, 0,50-0,95; p = 0,02) i 5-letnie przeżycie wolne od choroby (DFS disease free survival) okazało się statystycznie znamiennie dłuższe 34% vs 19% (HR dla progresji, 0,65; 95% CI, 0,48-0,89; p = 0,003). W analizie wielowariantowej dodatnimi czynnikami predykcyjnymi dla OS okazały się zastosowanie CTHO (p = 0,01) i lokalizacja guza w połączeniu przełykowo-żołądkowym (p < 0,01). Okołooperacyjna chemioterapia znamiennie zwiększyła resekcyjność z intencją wyleczenia (84% vs 73%; p = 0,04). Ciężkie objawy niepożądane związane z chemioterapią dotyczyły 38% chorych i były podobne do tych w badaniu MAGIC najczęściej występowała neutropenia (20,2%) [10]. Japońskie badanie randomizowane i prospektywne III fazy ACTS-GC [11] znacznie różniło się od badań europejskich. Zakładało bowiem, że jednorodną grupę chorych na operacyjnego GC w II (z wyjątkiem cechy T1) oraz IIIA i IIIB stopniu zaawansowania (zaawansowanie węzłowe wg klasyfikacji japońskiej), z cechami M0 i CY0 (ujemny wynik badania popłuczyn otrzewnowych) poddano radykalnej resekcji żołądka z limfadenektomią co najmniej D2, a następnie losowo przydzielono do obserwacji (ramię CH) lub do chemioterapii adiuwantowej (ramię CTHA) z użyciem leku S-1 (zawiera tegafur pochodną 5-FU oraz gimeracyl i oteracil, które hamują przemiany pirymidyn odpowiednio, dehydrogenazę dihydropirymidynową i fosforybozylotransferazę kwasu orotowego, w stosunku molowym 1:0,4:1). Analiza ITT objęła 1059 chorych. Po 3-latach ryzyko zgonu w ramieniu CTHA zmniejszyło się o 32% (HR, 0,68; 95% CI, 0,52 to 087; p = 0,003). Przeżycia 5-letnie, 32 33
18 całkowite (OS) i wolne od nawrotu (RFS relapse-fee survival), w całej grupie wyniosły odpowiednio: OS CTHA 71,7% vs CH 61,1%; HR, 0,669; 95% CI, 0, obniżenie ryzyka zgonu po zastosowaniu S-1 o 33,1%, RFS CTHA 65,4% vs CH 53,1%; HR, 0,653; 95% CI, 0,537-0,793 obniżenie ryzyka nawrotu GC po zastosowaniu S-1 o 34,7%. Bardziej szczegółowe dane z rozbiciem na podgrupy zaawansowania zostały przedstawione w tabeli I. Badanie ACTS-GC bezsprzecznie udowodniło korzyści dla przeżycia po podaniu adiuwantowej chemioterapii z użyciem pochodnej fluoropirymidynowej (S-1), choć z obowiązku należy wspomnieć, że japońska klasyfikacja zaawansowania GC różni się od AJCC/UICC, a także strategia operacyjna jest bardziej radykalna niż w Europie czy USA. Tym niemniej, przekładając zaawansowanie w badanej grupie na klasyfikację AJCC/UICC, 5-letni OS w ramionach CTHA vs CH uzyskało odpowiednio, 83,4% vs 70,8% (II), 68,9% vs 56,2% (IIIA), 43,7% vs 40,1% (IIIB) oraz 45,1% vs 42,7% (IV) chorych [11]. Uzyskane obniżenie względnego ryzyka zgonu w badaniu ACTS-GC było podobne do uzyskanych badaniach, amerykańskim SWOG 9008/INT 0116 [8] z adiuwantową chemioradioterapią oraz MAGIC i ACCORD-07 [9,10]. W tabeli II przestawiono porównanie wyników 3 badań (wyniki badania ACCORD-07 wypadły niemal identycznie, jak wyniki MAGIC, więc zrezygnowano z oddzielnego omówienia). Do badania ACTS-GC włączono jeden rodzaj operacyjnego pierwotnie nowotworu z potwierdzonym w badaniu histopatologicznym stadium zaawansowania. W pozostałych dwóch badaniach grupy były bardziej niejednorodne, gdyż oprócz GC włączono również pacjentów z EJC (INT0116 i MAGIC) oraz z EC (MAGIC), co spowodowało, że więcej było chorych z guzem T3/T4 w badaniach amerykańskim (69%) i europejskim (68%) w porównaniu z japońskim (46%). Z drugiej strony, potwierdzenie histopatologicznego zajęcia regionalnych węzłów chłonnych po operacji było w najmniejsze w badaniu MAGIC (74%), w porównaniu z INT0116 (85%) i ACTS-GC (89%) [8]. Tak więc, zaawansowanie nowotworów po operacji w tych badaniach miało różny zasięg największy guz MAGIC i INT0116, najwięcej węzłów ACTS-GC. Tym niemniej, niewątpliwy wpływ na uzyskanie najlepszych wyników przeżyć w badaniu ACTS-GC miały precyzyjne kryteria włączenia nowotworu poddanego operacji z intencją wyleczenia oraz najbardziej optymalny zakres limfadenektomii D2 u wszystkich chorych. W pozostałych dwóch badaniach zakres limfadenektomii był suboptymalny w INT0116 aż w około 90%, a w MAGIC trudno ustalić (nie podano dla EC), ale co najmniej w ponad 60% [8]. Można więc wysnuć wniosek, że gdyby obu tych badaniach wykonano optymalną limfadenektomię, znamienne różnice w przeżyciu na korzyść adiuwantowej chemioradioterapii vs CH (INT0116) lub okołooperacyjnej chemioterapii vs CH (MAGIC) prawdopodobnie by nie wystąpiły. Patrząc na opublikowane wyniki w/w badań (tabela 2), od razu widać, że różnica w przeżyciu pomiędzy ramieniem chirurgicznym w badaniu ACST-GC w porównaniu ramieniem z chemioradioterapią w INT0116 i ramieniem z chemioterapią okołooperacyjną w MAGIC była niemal dwukrotna na korzyść ACST-GC. Ponadto, ponad 1/3 chorych w badaniach INT0116 i MAGIC nie ukończyła zaplanowanego leczenia po operacji z powodu progresji raka, nietolerancji lub odmowy dalszego uczestnictwa. W styczniu 2015 r. autorzy koreańscy [12] opublikowali końcowe wyniki randomizowanego i prospektywnego badania III fazy ARTIST z losowym doborem chorych, porównującego adiuwantową chemioterapię (kapecytabina i cis platyna XP) z adiuwantową chemioradioterapią (XP + 45 Gy) u 458 chorych na operacyjnego raka żołądka (IB IV M0 wg AJCC), po optymalnej resekcji z limfadenektomią D2. Po 7 latach obserwacji prawdopodobieństwo przeżycia 5 lat wyniosło 73% w ramieniu CTHA i 75% w ramieniu CTH-RTH (p = 0,484), a DFS i OS (odpowiednio, HR, 0,740; 95% CI, 0,520-1,050; p = 0,0922 i HR, 1,130; 95% CI, 0,775-1,647; p = 0,5272) w obu ramionach nie różniły się istotnie. Po nawrocie raka przeżycie chorych do momentu zgonu było 9,7 miesięcy w ramieniu CTHA vs 7,2 miesięcy w ramieniu CTH-RTH (p = 0,076). W analizie podgrup zarysował się trend do poprawy DFS na korzyść ramienia z CTH-RTH u chorych z podtypem jelitowym GC (HR, 0,442; 0,231-0,845; p = 0,01) i z zajęciem węzłów chłonnych regionalnych (HR, 0,700; 0,493-0,994; p = 0,04), co znalazło przełożenie na mniejszą częstość nawrotów miejscowych (7% vs 13%; p = 0,0033), natomiast nie było różnic w odsetku przerzutów odległych między obu ramionami (24% vs 27%, p = 0,5568) [12]. Podsumowując część artykułu odnoszącą się do raka żołądka, należy stwierdzić, że chorzy na operacyjnego gruczolakoraka w II i III stopniu klinicznego zaawansowania wg AJCC powinni być leczeni za pomocą radykalnej resekcji (tj. z limfadenektomią D2) i adiuwantowej chemioterapii, zawierającej S-1 (populacja azjatycka) lub kapecytabinę i cis platynę (bez różnic populacyjnych). W przypadku zajęcia większej liczby węzłów chłonnych w materiale pooperacyjnym można rozważyć zastosowanie adiuwantowej chemioradioterapii. n Wnioski 1. Chorzy na płaskonabłonkowego raka przełyku, operacyjnego, o zaawansowaniu II-III wg AJCC powinni być leczeni za pomocą chemioradioterapii neoadiuwantowej z następową radykalną resekcją. 2. Chorzy na operacyjnego gruczolakoraka przełyku i połączenia przełykowo-żołądkowego o zaawansowaniu II-III wg AJCC powinni być leczeni za pomocą okołooperacyjnej chemioterapii z następową radykalną resekcją; alternatywnym sposobem przed operacją może być zastosowanie neoadiuwantowej chemioradioterapii. 3. Chorzy na operacyjnego gruczolakoraka żołądka o zaawansowaniu II-III i IV M0 wg AJCC powinni być leczeni za pomocą radykalnej resekcji z następową chemioterapią adiuwantową na bazie pochodnej fluoropirymidynowej z cis platyną. 4. U pacjentów z zaawansowaniem pn2/n3 w leczeniu adiuwantowym należy rozważyć zastosowanie chemioradioterapii zamiast chemioterapii. Piśmiennictwo 1. Wojciechowska U, Didkowska J, Zatoński W. Nowotwory złośliwe w Polsce w 2012 roku. Krajowy Rejestr Nowotworów. Ministerstwo Zdrowia, Warszawa 2014, Edge SB, Byrd D, Compton CC, et al. Esophagus and esophagogastric junction; stomach; exocrine and endocrine pancreas. AJCC Cancer Staging Handbook, 7th edition, Springer New York, 2010, ; ; Van Hagen P, Hulshof MCCM, van Lanschot JJB, et al. Preoperative chemoradiotherapy for esophageal and junctional cancer. N Engl J Med 2012, 366: Oppedijk V, van der Gaast A, van Lanschot JJB, et al. Patterns of recurrence after surgery alone versus preoperative chemoradiotherapy and surgery in the CROSS Trials. J Clin Oncol 2014, 32: Smyth EC, Waddell TS, Cunningham D. Optimal management of esophageal adenocarcinoma. Should we be CROSS? J Clin Oncol 2014, 32: Davies AR, Gossage JA, Zylstra J, et al. Tumor stage after neoadjuvant chemotherapy determines survival after surgery for adenocarcinoma of the esophagus and esophagogastric junction. J Clin Oncol 2014, 32: Stahl M, Walz MK, Stuschke M, et al. Phase III comparison of preoperative chemotherapy compared with chemoradiotherapy in patients with locally advanced adenocarcinoma of the esophagogastric junction. J Clin Oncol 2009, 27: Sano T. Adjuvant and neoadjuvant therapy of gastric cancer: a comparison of three pivotal studies. Curr Oncol Reports 2008, 10: Cunningham D, Allum WH, Stenning SP, et. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 2006, 355: Ychou M, Boige V, Pignon J-P, et al. Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol 2011, 29: Sasako M, Sakuramoto S, Katai H, et al. Five-year outcomes of a randomized phase III trial comparing adjuvant chemotherapy with S-1 versus surgery alone in stage II or III gastric cancer. J Clin Oncol 2011, 29: Park SH, Sohn TS, Lee J, et al. Phase III trial to compare adjuvant chemotherapy with capecitabine and cisplatin versus concurrent chemoradiotherapy in gastric cancer: final report of the adjuvant chemoradiotherapy in stomach tumors trial, including survival and subset analyses. J Clin Oncol 2015, January 5; DOI: /JCO
19 MANAGEMENT NOWOTWORY GÓRNEGO ODCINKA UKŁADU POKARMOWEGO: PROBLEM CHIRURGICZNY CZY ONKOLOGICZNY? ANDRZEJ W. SZAWŁOWSKI Centrum Onkologii Instytut im. Marii Skłodowskiej-Curie w Warszawie Adres korespondecyjny: Warszawa, ul. Wawelska 15B tel Do złośliwych nowotworów górnego odcinka układu pokarmowego (GOUP) zalicza się: rak przełyku, rak żołądka, rak trzustki, rak dróg żółciowych zewnątrzwątrobowych (w tym rak brodawki Vatera) i rak wątroby. Należy do n ich również rak dwunastnicy, ale występuje on rzadko. Granicą między górnym a dolnym odcinkiem przewodu pokarmowego (część rurowa układu pokarmowego) jest więzadło Treitza. Według ostatnio opublikowanych danych przez Krajowy Rejestr Nowotworów w Polsce [1] rak żołądka, najczęstszy z nowotworów GOUP, znajduje się na 6-tym miejscu u mężczyzn i na 10-tym miejscu u kobiet w kolejności zarejestrowanej zachorowalności na złośliwe nowotwory (3399 i 1877 nowych zachorowań, odpowiednio), rak trzustki na 10-tym miejscu u mężczyzn i 12-tym miejscu u kobiet (1626 i 1628 nowych zachorowań, odpowiednio), rak przełyku na 15-tym miejscu u mężczyzn i 35-tym u kobiet (929 i 243 nowych zachorowań, odpowiednio), rak wątroby na 17-tym miejscu u mężczyzn i 23-tym u kobiet (829 i 587 nowych zachorowań, odpowiednio) i rak pęcherzyka żółciowego (główny przedstawiciel raka dróg żółciowych zewnątrzwątrobowych) na 38 miejscu u mężczyzn i 17-tym u kobiet (180 i 795 nowych zachorowań, odpowiednio). Problem kliniczny W Polsce i w krajach Unii Europejskiej problem zaawansowanych złośliwych nowotworów GOUP charakteryzuje fakt, że jest więcej naukowych publikacji na temat tych nowotworów niż chorych przeżywających 5 lat po leczeniu (zasadnicze kryterium w onkologii skuteczności leczenia). Wynika to z faktu, że nie ma badań przesiewowych (skryningowych) służących wykryciu przypadków wczesnych, które w tych rozpoznaniach diagnozowane są rzadko (zwykle przypadkowo), stopniowanie czyli ustalenie klinicznego stopnia zaawansowania, co określa możliwości leczenia jest zwykle mało precyzyjne, resekcyjność/operacyjność dotyczy niewielu chorych (wyjątek: rak żołądka), a potencjalne powikłania związane z leczeniem mogą stanowić zagrożenie życia. W związku z tym, w porównaniu z innymi nowotworami (na przykład: rak piersi czy rak jelita grubego) w odniesieniu do nowotworów GOUP, obserwuje się niekorzystne farmakoekonomiczne współczynniki: koszt korzyść i ryzyko korzyść, co sprawia, że niewiele ośrodków podejmuje się leczenia tych nowotworów. Nowotwory GOUP należą do litych nowotworów i w tej grupie chirurgia była, jest i na pewno przez długi czas jeszcze będzie, tak zwanym złoty standardem leczenia. W przypadku raka płaskonabłonkowego przełyku możliwe jest też skuteczne zastosowanie radioterapii (nowotwór promieniowrażliwy) [2]. W pozostałych przypadkach radio- i/lub chemioterapia wchodzą w skład skojarzonego leczenia jako leczenie przedoperacyjne (neo-adiuwantowe, indukcyjne) lub pooperacyjne (adiuwantowe) albo paliatywne. Stopień klinicznego zaawansowania a możliwości chirurgicznego leczenia Według zbiorczych danych z różnych grup narządowych złośliwych nowotworów, w tym nowotworów GOUP wiadomo, że [3]: Stopień I (T1, NO, MO), czyli zaawansowanie miejscowe i nowotwór ograniczony do jednego narządu bez jego przekraczania: wyleczenie %; Stopień II (T1-2, N1, MO), czyli zaawansowanie loko- -regionalne, to znaczy nowotwór zajmuje narząd i daje przerzuty do regionalnych węzłów chłonnych: szansa na wyleczenie około 50%; Stopień III (T1-3, No-2, MO), czyli rozległe zajęcie loko- -regionalne: szansa na wyleczenie około 20%; Stopień IV (każde T, każde N, M1), czyli nowotwór zaawansowany z odległymi przerzutami: szansa na wyleczenie do 5%. Do leczenia z intencją wyleczenia (radykalnego) kwalifikują się zatem chorzy w I-III stopniu klinicznego zaawansowania (T1-4, NO-3, MO), zaś do paliatywnego leczenia chorzy w IV-tym stopniu klinicznego zaawansowania (T1-4, No-3, M1). Z danych tych wynika, że idealnymi kandydatami do wyłącznie chirurgicznego leczenia, w tym metodami chirurgii mało inwazyjnej (na przykład: chirurgia endoskopowa) Chorzy resekcyjni/operacyjni Neo-adiuwant Guz: T1-T3 Resekcyjność (downstaging) Indukcja regresji Guz: T4 Chorzy nieresekcyjni CHIRURGIA Rycina 1. TAKTYKA SKOJARZONEGO LECZENIA NOWOTWORÓW GÓRNEGO ODCINKA UKŁADU POKARMOWEGO Tabela I. SKUTECZNOŚĆ CHIRURGICZNEGO LECZENIA ZŁOŚLIWYCH NOWOTWORÓW GOUP W POLSCE (4) Rak Rak Rak Rak dróg żółciowych Rak wątroby przełyku żołądka trzustki zewnątrzwątrobowych Pierwotny Wtórny* Resekcyjność/Operacyjność 10% 50-70% 15-20% 20% 15-20% 15-20% Przeżycie 5-letnie Pojedynczy: 5% 25% 6-7% < 5% 0-20% Pojedyncze 30-40% przypadki Mnogi: * przerzuty raka jelita grubego wątroby są chorzy w I-szym stopniu klinicznego zaawansowania. Natomiast chorzy o zaawansowaniu II/III czyli loko-regionalnym dla poprawy wyników chirurgicznego leczenia wymagają postępowania skojarzonego (chirurgia + chemioterapia + radioterapia). Skuteczność chirurgicznego leczenia zaawansowanych złośliwych nowotworów GOUP w Polsce przedstawiono zbiorczo w tabeli 1. Podsumowanie Nowotwory GOUP są w przeważającej większości przypadków diagnozowane w zaawansowanych stadiach rozwoju co rzutuje na możliwości skutecznego leczenia. Dodatkowo są to nowotwory o złej biologii co sprawia, że nawet w doświadczonych ośrodkach wyniki chirurgicznego leczenia są niezadowalające. Dlatego w świetle aktualnego stanu wiedzy nowotwory GOUP pozostają głównie nadal nierozwiązanym problemem onkologicznym. W przypadku właściwej kwalifikacji do chirurgicznego leczenia są szanse na uzyskanie dobrych wyników 5-letnich przeżyć choć nie zawsze towarzyszy temu dobra jakość życia. Szansa na poprawę wyników chirurgicznych w zaawansowanych stadiach nowotworu należy upatrywać w leczeniu skojarzonym, głównie neo-adiuwantowym i/lub adiuwantowym. n Chirurgia radykalna (resekcja: R0) Adiuwant Paliacja Chirurgia nieradykalna (resekcja: R1-R2) Chemioterapia pt1-3 N1-3 M0 Radykalizm systemowy (przerzuty) Radioterapia pt3/4 N0-3 M0 Radykalizm lokalny (wznowa) Leczenie objawowe (terminalne) RO operacja makro- i mikroskopowo radykalna R1 operacja mikroskopowo nieradykalna R2 operacja makroskopowo nieradykalna Piśmiennictwo 1. Wojciechowska U., Didkowska J., Zatoński W. Nowotwory złośliwe w Polsce w 2010 roku. Wyd. Centrum Onkologii-Instytut im Marii Skłodowskiej-Curfie w Warszawie, 2012 r. 2. Szawłowski A.W. Nowotwory przełyku. W: A.Jeziorski, A.W.Szawłowski, E.Towpik (red.) Chirurgia onkologiczna, Wyd. Wydawnictwo Lekarskie PZWL, Warszawa, 2009 ; tom IV: Szawłowski A.W. Zasady rozpoznawania i leczenia nowotworów złośliwych. W: W. Noszczyk (red.) Chirurgia, Wyd. Wydawnictwo Lekarskie PZWL, Warszawa, 2005 ; tom I: Szawłowski A.W., Jeziorski K. Nowotwory złosliwe górnego odcinka układu pokarmowego. W: M. Krzakowski ( red.) Onkologia klioniczna, Wyd. Borgis, Warszawa, 2001; tom II:
20 OCENA PATOMORFOLOGICZNA NOWOTWORÓW GÓRNEGO ODCINKA PRZEWODU POKARMOWEGO ANNA NASIEROWSKA-GUTTMEJER Zakład Patomorfologii CSK MSW w Warszawie Kierownik Zakładu: prof. dr hab. med. Anna Nasierowska-Guttmejer Adres do korespondencji: Prof. dr hab. med. Anna Nasierowska- Guttmejer Zakład Patomorfologii CSK MSW w Warszawie ul. Wołoska 137, Warszawa anna.nasierowska@cskmswia.pl Raport patomorfologiczny Nowe metody leczenia nowotworów górnego odcinka przewodu pokarmowego, jak miejscowe leczenie endoskopowe, leczenie przedoperacyjne w przypadkach resekcyjnych zaawansowanych miejscowo, ulepszanie technik operacyjnych oraz terapia celowana molekularnie wymagają od patomorfologów aktualizacji zasad diagnostyki i dostosowywania ich do kliniki. Podstawową formą komunikacji między patomorfologami, a klinicystami jest raport patomorfologiczny. Zawiera on cechy makro- i mikroskopowe nowotworu mające istotne znaczenie dla planowania właściwego leczenia i określenia rokowania chorych. Dostarcza on informacji przydatnych klinicznie na temat umiejscowienia, typu, stopnia dojrzałości i za awansowania oraz innych swoistych narządowo cech mikroskopowych nowotworu z uwzględnieniem czynników prognostycznych i predykcyjnych badanych metodami immunohistochemicznymi i molekularnymi. Dlatego tak ważne jest, aby patomorfolodzy wzorowali się na ujednoliconej formie raportu patomorfologicznego, który powinien spełniać cztery cechy: terminowość, kompletność, dokładność i użyteczność kliniczną. Polskie Towarzystwo Patologów wychodząc naprzeciw wymienionym wskazaniom opracowało Zalecenia do diagnostyki histopatologicznej nowotworów oparte na raportach synoptycznych Kollegium Amerykańskiego Towarzystwa Patologów (College of Streszczenie: Nowotwory w górnym odcinku układu pokarmowego powstają w przełyku, okolicy połączenia przełykowo-żołądkowego i żołądku. Pod względem typu histologicznego wyróżnia się raki płaskonabłonkowego (powstające głównie w przełyku), raki gruczołowe (rzadko w przełyku, głównie w dolnym jego odcinku, okolicy połączenia przełykowo-żołądkowego i w żołądku), nowotwory neuroendokrynne i nowotwory podścieliskowe (GIST). Stopień zaawansowania raka gruczołowego i płaskonabłonkowego (ptnm), poza jego typem histologicznym i stopniem dojrzałości (G) jest istotnym czynnikiem rokowniczym. W nowotworach neuroendokrynnych czynnikiem prognostycznym i predykcyjnym jest stopień jego zróżnicowania i indeks proliferacyjny Ki67 określający stopień dojrzałości nowotworu (G). Wymienione parametry dzielą ten typ nowotworu na wysokozróżnicowane (NEN) i raki neuroendokrynne (NEC). Czynnikami prognostycznymi i predykcyjnymi w GIST jest lokalizacja nowotworu (żołądek lub jelito cienkie i grube), wielkość guza, liczba figur podziału w 50 dpw oraz miejsce mutacji w genie C-KIT. Receptor HER2 jest czynnikiem predykcyjnym w odniesieniu do leczenia Trastuzumabem raka żołądka. Jego ocena znajduje zastosowanie w raku gruczołowym połączenia przełykowożołądkowego w 24 do 34% przypadków i raka żołądka, częściej odcinka proksymalnego, niż dystalnego. Podstawową formą komunikacji między patomorfologami, a klinicystami jest raport patomorfologiczny, który zawiera informacji przydatnych klinicznie na temat umiejscowienia, typu, stopnia dojrzałości i za awansowania oraz innych swoistych narządowo cech mi kroskopowych nowotworu z uwzględnieniem czynników prognostycznych i predykcyjnych badanych metodami immunohistochemicznymi i molekularnymi. Słowa kluczowe: Rak płaskonabłonkowy przełyku, rak połączenia przełykowo-żołądkowego, rak gruczołowy żołądka, HER2 American Pathologists). Podział anatomiczny Anatomiczny podział górnego odcinka przewodu pokarmowego uwzględnia przełyk, okolicę połączenia przełykowo-żołądkowego i żołądek. Dla raków okolicy połączenia przełykowo-żołądkowego istnieją definicje kliniczne, jak endoskopowa i chirurgiczna według Sieverta z podziałem na I, II i III typ. Według klasyfikacji Światowej Organizacji Zdrowia WHO z 2009 roku raki gruczołowe naciekające połączenie przełykowo-żołądkowe, niezależnie od umiejscowienia ich głównej masy, określane są terminem raka połączenia przełykowo-żołądkowego (adenocarcinoma of the esophagogastric junction, OGJ). Natomiast rak gruczołowy zlokalizowany w dolnym odcinku przełyku, ponad połączeniem przełykowo-żołądkowym i nie naciek PPŻ, jest rakiem gruczołowym przełyku. Rak gruczołowy bliższej części żołądka nie naciekający połączenia przełyko-żołądkowego definiowany jest jako rak pochodzenia żołądkowego, mimo opinii kontrowersyjnych, iż może pochodzić z pluripotencjalnych komórek nabłonkowych okolicy połączenia przełykowo-żołądkowego, określany jest coraz częściej terminem raka bliższej części żołądka (carcinoma of the proximal stomach), który jest lepszy od niejednoznacznego stosowanego również określenia rak wpustu. Typ histologiczny Nowotwory górnego odcinka przewodu pokarmowego dzielone są w zależności od ich histogenezy na nowotwory pochodzenia nabłonkowego, głównie raki płaskonabłonkowe i raki gruczołowe oraz nowotwory neuroendokrynne i mezenchymalne. Przełyk. W przełyku dominują raki płaskonabłonkowe (95%) przypadków. Raki gruczołowe stanowią poniżej 5% przypadków, zlokalizowane są przeważnie w dolnym odcinku przełyku i rozwijają się na podłożu metaplazji Barrett a. Do rzadkości należy ich umiejscowienie w odcinku górnym narządu i występowanie w terenie ektopii błony śluzowej żołądka. Sporadycznie występują warianty raka płaskonabłonkowego jak raki wrzecionowatokomórkowe (metaplastyczne) i brodawczakowate (verrucous carcinoma) oraz nowotwory typu śliniankowego i czerniaki. Również rzadko rozpoznawane są nowotwory mezenchymalne jak mięśniaki, naczyniaki, guz z komórek ziarnistych (granular cell tumor), tłuszczaki lub nowotwory podścieliskowe, GIST. W zależności od stopnia dojrzałości raki płaskonabłonkowe i gruczołowe dzielone są na wysokodojrzałe (G1), średniodojrzałe (G2) i niskodojrzałe (G3). W przypadku raka płaskonabłonkowego określa się go na podstawie obszaru o niskiej dojrzałości. W przypadku raka gruczołowego uwzględnia się powierzchnię zajętą przez cewy gruczołowe (G1 >95% utkania, G %, G3 <49%). Dla uproszczenia raki niezróżnicowane (niewykazujące różnicowania ani w kierunku płaskonabłonkowym ani gruczołowym), dawniej klasyfikowane jako G4, obecnie zostały włączone do grupy G3 raków płaskonabłonkowych przełyku. Połączenie przełykowo-żołądkowe (PPŻ). Do raków tej lokalizacji zaliczane są przypadki, które według wcześniejszej klasyfikacji WHO określane były terminem rak wpustu. W okolicy połączenia przełykowo-żołądkowego dominują raki gruczołowe, które powstają na podłożu metaplazji jelitowej typu przełyku Barrett a lub zapalenia zanikowego błony śluzowej proksymalnej części żołądka typu wpustu lub trzonu żołądka i naciekają okolice PPŻ. Spotykane są również raki płaskonabłonkowe szerzące się z dolnego odcinka przełyku do PPŻ. Żołądek. Uwzględniając histogenezę nowotworów, do najczęstszych nowotworów żołądka należą raki gruczołowe i nowotwory neuroendokrynne, a wśród mezenchymalnych - nowotwory podścieliskowe (GIST). A. Raki żołądka ocenia się według obowiązującej klasyfikacji WHO z 2010 roku, która wyróżnia następujące ich typy: Rak gruczołowy (ang. adenocarcinoma) Rak gruczołowy brodawkowaty (ang. papillary adenocarcinoma) Rak gruczołowy cewkowy (ang. tubular adenocarcinoma) Rak gruczołowy śluzowy (ang. mucinous adenocarcinoma) Rak o małej spoistości w tym rak śluzowo komórkowy i inne warianty (ang. poorly cohesive carcinoma including signet ring cell carcinoma and other variants) Rak gruczołowy mieszany (ang. mixed adenocarcinoma) Rak gruczołowo płaskonabłonkowy (ang. adenosqumous carcinoma) Rak z naciekiem limfocytarnym w podścielisku (rak rdzeniasty) [ang. carcinoma with lymphoid stroma (medullary carcinoma)] Rak gruczołowy podobny do wątrobowo komórkowego (ang. hepatoid adenocarcinoma) Rak płaskonabłonkowy (ang. squamous cell carcinoma) Rak neuroendokrynny (ang. Carcinoma neuroendocrinale, NEC) z dużej komórki (ang. Large cell NEC) z małej komórki (ang. Small cell NEC) Rak niezróżnicowany (ang. undifferentiated carcinoma) Alternatywną klasyfikacją histologiczną raków żołądka jest podział według Laurena, który uwzględnia: Typ I jelitowy, ograniczony zawierający w swoim utkaniu głównie elementy raka gruczołowego w różnym stopniu zróżnicowania, Typ II rozlany zawierający w swoim utkaniu powyżej 50% raka o małej spoistości w tym raka śluzowo komórkowego i inne jego warianty Typ III postać mieszana zawierająca w swoim utkaniu zarówno elementy raka gruczołowego w różnym stopniu zróżnicowania jak i raka o małej spoistości?? w tym raka śluzowo komórkowy i inne jego warianty. B. Nowotwory neuroendokrynne (NET guzy neuroendokrynne, NEN nowotwory neuroendokrynne) stanowią heterogenna grupę nowotworów rozwijających się z rozlanego systemu endokrynnego (wewnątrzwydzielniczego). Typ komórki i umiejscowienie guza decydują o przebiegu klinicznym i ich obrazie histopatologicznym. Kolejne zmiany w klasyfikacji WHO nowotworów neuroendokrynnych przedstawiono w Tabeli I. W 2010 roku, został przyjęty nowy podział NET na dwie podstawowe grupy: nowotwory wysoko i niskozróżnicowane. Opierał się on po pierwsze na ocenie morfologii guza, której punktem odniesienia były struktury tworzone przez komórki nienowotworowe. Drugim parametrem był stopień dojrzałości (G - grading) guza, który odzwierciedlał potencjalny przebieg kliniczny nowotworu. Istotną różnicą w porównaniu do klasyfikacji z 2000 roku było po pierwsze nazwanie wysokozróżnicowanych guzów i wysokozróżnicowanych raków jednym terminem guzów/ nowotworów neuroendokrynnych (NEN), natomiast cecha G decydowała o ich stopniu dojrzałości (G1 lub G2). Po drugie, niskozróżnicowane raki neuroendokrynne drobno- lub wielkokomórkowych (G3) określono nazwą raka neuroendokrynnego (neuroendocrine carcinoma NEC ) traktując je według kryteriów diagnostycznych i terapii jak klasyczne raki. Tabele I i III przedstawiają klasyfikacje WHO NEN w 1980 roku, 2000 roku i 2010 roku, a Tabela II kryteria oceny stopnia histologicznej dojrzałości nowotworów neuroendokrynnych (cecha G). NEN żołądka zwykle są nieczynnymi hormonalnie guzami z komórek ECL (enterochromaffin-like) produkujących histaminę, które przeważnie występują w trzonie i dnie żołądka. Rzadziej spotykanymi są liczne w odźwierniku 38 39
SSW1.1, HFW Fry #20, Zeno #25 Benchmark: Qtr.1. Fry #65, Zeno #67. like
 SSW1.1, HFW Fry #20, Zeno #25 Benchmark: Qtr.1 I SSW1.1, HFW Fry #65, Zeno #67 Benchmark: Qtr.1 like SSW1.2, HFW Fry #47, Zeno #59 Benchmark: Qtr.1 do SSW1.2, HFW Fry #5, Zeno #4 Benchmark: Qtr.1 to SSW1.2,
SSW1.1, HFW Fry #20, Zeno #25 Benchmark: Qtr.1 I SSW1.1, HFW Fry #65, Zeno #67 Benchmark: Qtr.1 like SSW1.2, HFW Fry #47, Zeno #59 Benchmark: Qtr.1 do SSW1.2, HFW Fry #5, Zeno #4 Benchmark: Qtr.1 to SSW1.2,
 aforementioned device she also has to estimate the time when the patients need the infusion to be replaced and/or disconnected. Meanwhile, however, she must cope with many other tasks. If the department
aforementioned device she also has to estimate the time when the patients need the infusion to be replaced and/or disconnected. Meanwhile, however, she must cope with many other tasks. If the department
Proposal of thesis topic for mgr in. (MSE) programme in Telecommunications and Computer Science
 Proposal of thesis topic for mgr in (MSE) programme 1 Topic: Monte Carlo Method used for a prognosis of a selected technological process 2 Supervisor: Dr in Małgorzata Langer 3 Auxiliary supervisor: 4
Proposal of thesis topic for mgr in (MSE) programme 1 Topic: Monte Carlo Method used for a prognosis of a selected technological process 2 Supervisor: Dr in Małgorzata Langer 3 Auxiliary supervisor: 4
Analiza skuteczności i bezpieczeństwa leczenia systemowego najczęściej występujących nowotworów
 Uniwersytet Medyczny w Lublinie Rozprawa doktorska streszczenie rozprawy doktorskiej Analiza skuteczności i bezpieczeństwa leczenia systemowego najczęściej występujących nowotworów u chorych w podeszłym
Uniwersytet Medyczny w Lublinie Rozprawa doktorska streszczenie rozprawy doktorskiej Analiza skuteczności i bezpieczeństwa leczenia systemowego najczęściej występujących nowotworów u chorych w podeszłym
Udział czynników demograficznych w kształtowaniu wyników leczenia raka jajnika na podstawie materiału Gdyńskiego Centrum Onkologii
 Lek. med. Andrzej Kmieć Udział czynników demograficznych w kształtowaniu wyników leczenia raka jajnika na podstawie materiału Gdyńskiego Centrum Onkologii Rozprawa na stopień doktora nauk medycznych Zakład
Lek. med. Andrzej Kmieć Udział czynników demograficznych w kształtowaniu wyników leczenia raka jajnika na podstawie materiału Gdyńskiego Centrum Onkologii Rozprawa na stopień doktora nauk medycznych Zakład
Tychy, plan miasta: Skala 1: (Polish Edition)
 Tychy, plan miasta: Skala 1:20 000 (Polish Edition) Poland) Przedsiebiorstwo Geodezyjno-Kartograficzne (Katowice Click here if your download doesn"t start automatically Tychy, plan miasta: Skala 1:20 000
Tychy, plan miasta: Skala 1:20 000 (Polish Edition) Poland) Przedsiebiorstwo Geodezyjno-Kartograficzne (Katowice Click here if your download doesn"t start automatically Tychy, plan miasta: Skala 1:20 000
Radiologiczna ocena progresji zmian próchnicowych po zastosowaniu infiltracji. żywicą o niskiej lepkości (Icon). Badania in vivo.
 Renata Zielińska Radiologiczna ocena progresji zmian próchnicowych po zastosowaniu infiltracji żywicą o niskiej lepkości (Icon). Badania in vivo. Rozprawa na stopień doktora nauk medycznych Zakład Stomatologii
Renata Zielińska Radiologiczna ocena progresji zmian próchnicowych po zastosowaniu infiltracji żywicą o niskiej lepkości (Icon). Badania in vivo. Rozprawa na stopień doktora nauk medycznych Zakład Stomatologii
Uniwersytet Medyczny w Łodzi. Wydział Lekarski. Jarosław Woźniak. Rozprawa doktorska
 Uniwersytet Medyczny w Łodzi Wydział Lekarski Jarosław Woźniak Rozprawa doktorska Ocena funkcji stawu skokowego po leczeniu operacyjnym złamań kostek goleni z uszkodzeniem więzozrostu piszczelowo-strzałkowego
Uniwersytet Medyczny w Łodzi Wydział Lekarski Jarosław Woźniak Rozprawa doktorska Ocena funkcji stawu skokowego po leczeniu operacyjnym złamań kostek goleni z uszkodzeniem więzozrostu piszczelowo-strzałkowego
Zakopane, plan miasta: Skala ok. 1: = City map (Polish Edition)
 Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Click here if your download doesn"t start automatically Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Zakopane,
Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Click here if your download doesn"t start automatically Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Zakopane,
Analysis of infectious complications inf children with acute lymphoblastic leukemia treated in Voivodship Children's Hospital in Olsztyn
 Analiza powikłań infekcyjnych u dzieci z ostrą białaczką limfoblastyczną leczonych w Wojewódzkim Specjalistycznym Szpitalu Dziecięcym w Olsztynie Analysis of infectious complications inf children with
Analiza powikłań infekcyjnych u dzieci z ostrą białaczką limfoblastyczną leczonych w Wojewódzkim Specjalistycznym Szpitalu Dziecięcym w Olsztynie Analysis of infectious complications inf children with
Unit of Social Gerontology, Institute of Labour and Social Studies ageing and its consequences for society
 Prof. Piotr Bledowski, Ph.D. Institute of Social Economy, Warsaw School of Economics local policy, social security, labour market Unit of Social Gerontology, Institute of Labour and Social Studies ageing
Prof. Piotr Bledowski, Ph.D. Institute of Social Economy, Warsaw School of Economics local policy, social security, labour market Unit of Social Gerontology, Institute of Labour and Social Studies ageing
Warsztaty Ocena wiarygodności badania z randomizacją
 Warsztaty Ocena wiarygodności badania z randomizacją Ocena wiarygodności badania z randomizacją Każda grupa Wspólnie omawia odpowiedź na zadane pytanie Wybiera przedstawiciela, który w imieniu grupy przedstawia
Warsztaty Ocena wiarygodności badania z randomizacją Ocena wiarygodności badania z randomizacją Każda grupa Wspólnie omawia odpowiedź na zadane pytanie Wybiera przedstawiciela, który w imieniu grupy przedstawia
Ocena częstości występowania bólów głowy u osób chorych na padaczkę.
 lek. med. Ewa Czapińska-Ciepiela Ocena częstości występowania bólów głowy u osób chorych na padaczkę. Rozprawa na stopień doktora nauk medycznych Promotor Prof. dr hab. n. med. Jan Kochanowski II Wydział
lek. med. Ewa Czapińska-Ciepiela Ocena częstości występowania bólów głowy u osób chorych na padaczkę. Rozprawa na stopień doktora nauk medycznych Promotor Prof. dr hab. n. med. Jan Kochanowski II Wydział
WPŁYW AKTYWNOŚCI FIZYCZNEJ NA STAN FUNKCJONALNY KOBIET PO 65 ROKU ŻYCIA Z OSTEOPOROZĄ
 WPŁYW AKTYWNOŚCI FIZYCZNEJ NA STAN FUNKCJONALNY KOBIET PO 65 ROKU ŻYCIA Z OSTEOPOROZĄ Streszczenie: Wstęp: Proces starzenia się w naturalny sposób wpływa na ograniczenie sprawności, nawet w sytuacji zachowania
WPŁYW AKTYWNOŚCI FIZYCZNEJ NA STAN FUNKCJONALNY KOBIET PO 65 ROKU ŻYCIA Z OSTEOPOROZĄ Streszczenie: Wstęp: Proces starzenia się w naturalny sposób wpływa na ograniczenie sprawności, nawet w sytuacji zachowania
Urszula Coupland. Zaburzenia neurologiczne u dzieci wertykalnie zakażonych HIV. Rozprawa na stopień doktora nauk medycznych
 Urszula Coupland Zaburzenia neurologiczne u dzieci wertykalnie zakażonych HIV Rozprawa na stopień doktora nauk medycznych Klinika Chorób Zakaźnych Wieku Dziecięcego Warszawskiego Uniwersytetu Medycznego
Urszula Coupland Zaburzenia neurologiczne u dzieci wertykalnie zakażonych HIV Rozprawa na stopień doktora nauk medycznych Klinika Chorób Zakaźnych Wieku Dziecięcego Warszawskiego Uniwersytetu Medycznego
Evaluation of the main goal and specific objectives of the Human Capital Operational Programme
 Pracownia Naukowo-Edukacyjna Evaluation of the main goal and specific objectives of the Human Capital Operational Programme and the contribution by ESF funds towards the results achieved within specific
Pracownia Naukowo-Edukacyjna Evaluation of the main goal and specific objectives of the Human Capital Operational Programme and the contribution by ESF funds towards the results achieved within specific
Charakterystyka kliniczna chorych na raka jelita grubego
 Lek. Łukasz Głogowski Charakterystyka kliniczna chorych na raka jelita grubego Rozprawa na stopień doktora nauk medycznych Opiekun naukowy: Dr hab. n. med. Ewa Nowakowska-Zajdel Zakład Profilaktyki Chorób
Lek. Łukasz Głogowski Charakterystyka kliniczna chorych na raka jelita grubego Rozprawa na stopień doktora nauk medycznych Opiekun naukowy: Dr hab. n. med. Ewa Nowakowska-Zajdel Zakład Profilaktyki Chorób
ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA JEZYKOWA) BY DOUGLAS KENT HALL
 Read Online and Download Ebook ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA JEZYKOWA) BY DOUGLAS KENT HALL DOWNLOAD EBOOK : ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA Click link bellow and free register
Read Online and Download Ebook ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA JEZYKOWA) BY DOUGLAS KENT HALL DOWNLOAD EBOOK : ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA Click link bellow and free register
Effective Governance of Education at the Local Level
 Effective Governance of Education at the Local Level Opening presentation at joint Polish Ministry OECD conference April 16, 2012, Warsaw Mirosław Sielatycki Ministry of National Education Doskonalenie
Effective Governance of Education at the Local Level Opening presentation at joint Polish Ministry OECD conference April 16, 2012, Warsaw Mirosław Sielatycki Ministry of National Education Doskonalenie
Justyna Kinga Stępkowska
 Warszawski Uniwersytet Medyczny Wydział Nauki o Zdrowiu Justyna Kinga Stępkowska Ilościowa ocena wybranych antygenów nowotworowych u zdrowych kobiet stosujących dwuskładnikową antykoncepcję hormonalną
Warszawski Uniwersytet Medyczny Wydział Nauki o Zdrowiu Justyna Kinga Stępkowska Ilościowa ocena wybranych antygenów nowotworowych u zdrowych kobiet stosujących dwuskładnikową antykoncepcję hormonalną
Pułapki z pozycji radioterapeuty GLEJAKI. dr n. med. Milena Szacht Centrum Radioterapii CSK MSWiA w Warszawie
 Pułapki z pozycji radioterapeuty GLEJAKI dr n. med. Milena Szacht Centrum Radioterapii CSK MSWiA w Warszawie Pułapka 1 błędne przekonanie o dobrej skuteczności medycyny w leczeniu glejaków Jestem dobrym
Pułapki z pozycji radioterapeuty GLEJAKI dr n. med. Milena Szacht Centrum Radioterapii CSK MSWiA w Warszawie Pułapka 1 błędne przekonanie o dobrej skuteczności medycyny w leczeniu glejaków Jestem dobrym
Cracow University of Economics Poland. Overview. Sources of Real GDP per Capita Growth: Polish Regional-Macroeconomic Dimensions 2000-2005
 Cracow University of Economics Sources of Real GDP per Capita Growth: Polish Regional-Macroeconomic Dimensions 2000-2005 - Key Note Speech - Presented by: Dr. David Clowes The Growth Research Unit CE Europe
Cracow University of Economics Sources of Real GDP per Capita Growth: Polish Regional-Macroeconomic Dimensions 2000-2005 - Key Note Speech - Presented by: Dr. David Clowes The Growth Research Unit CE Europe
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Clinical Trials. Anna Dziąg, MD, ąg,, Associate Director Site Start Up Quintiles
 Polandchallenges in Clinical Trials Anna Dziąg, MD, ąg,, Associate Director Site Start Up Quintiles Poland- legislation 1996-2003 Ustawa z dnia 5 grudnia 1996 r o zawodach lekarza i lekarza dentysty, z
Polandchallenges in Clinical Trials Anna Dziąg, MD, ąg,, Associate Director Site Start Up Quintiles Poland- legislation 1996-2003 Ustawa z dnia 5 grudnia 1996 r o zawodach lekarza i lekarza dentysty, z
MaPlan Sp. z O.O. Click here if your download doesn"t start automatically
 Mierzeja Wislana, mapa turystyczna 1:50 000: Mikoszewo, Jantar, Stegna, Sztutowo, Katy Rybackie, Przebrno, Krynica Morska, Piaski, Frombork =... = Carte touristique (Polish Edition) MaPlan Sp. z O.O Click
Mierzeja Wislana, mapa turystyczna 1:50 000: Mikoszewo, Jantar, Stegna, Sztutowo, Katy Rybackie, Przebrno, Krynica Morska, Piaski, Frombork =... = Carte touristique (Polish Edition) MaPlan Sp. z O.O Click
ERASMUS + : Trail of extinct and active volcanoes, earthquakes through Europe. SURVEY TO STUDENTS.
 ERASMUS + : Trail of extinct and active volcanoes, earthquakes through Europe. SURVEY TO STUDENTS. Strona 1 1. Please give one answer. I am: Students involved in project 69% 18 Student not involved in
ERASMUS + : Trail of extinct and active volcanoes, earthquakes through Europe. SURVEY TO STUDENTS. Strona 1 1. Please give one answer. I am: Students involved in project 69% 18 Student not involved in
Ingrid Wenzel. Rozprawa doktorska. Promotor: dr hab. med. Dorota Dworakowska
 Ingrid Wenzel KLINICZNE ZNACZENIE EKSPRESJI RECEPTORA ESTROGENOWEGO, PROGESTERONOWEGO I ANDROGENOWEGO U CHORYCH PODDANYCH LECZNICZEMU ZABIEGOWI OPERACYJNEMU Z POWODU NIEDROBNOKOMÓRKOWEGO RAKA PŁUC (NDKRP)
Ingrid Wenzel KLINICZNE ZNACZENIE EKSPRESJI RECEPTORA ESTROGENOWEGO, PROGESTERONOWEGO I ANDROGENOWEGO U CHORYCH PODDANYCH LECZNICZEMU ZABIEGOWI OPERACYJNEMU Z POWODU NIEDROBNOKOMÓRKOWEGO RAKA PŁUC (NDKRP)
Ocena przydatności rezonansu magnetycznego w określaniu czasu wystąpienia wczesnego
 STRESZCZENIE: Ocena przydatności rezonansu magnetycznego w określaniu czasu wystąpienia wczesnego udaru niedokrwiennego mózgu. Udar jest drugą co do częstości przyczyną zgonów na Świecie i podstawową przyczyną
STRESZCZENIE: Ocena przydatności rezonansu magnetycznego w określaniu czasu wystąpienia wczesnego udaru niedokrwiennego mózgu. Udar jest drugą co do częstości przyczyną zgonów na Świecie i podstawową przyczyną
Rozpoznawanie twarzy metodą PCA Michał Bereta 1. Testowanie statystycznej istotności różnic między jakością klasyfikatorów
 Rozpoznawanie twarzy metodą PCA Michał Bereta www.michalbereta.pl 1. Testowanie statystycznej istotności różnic między jakością klasyfikatorów Wiemy, że możemy porównywad klasyfikatory np. za pomocą kroswalidacji.
Rozpoznawanie twarzy metodą PCA Michał Bereta www.michalbereta.pl 1. Testowanie statystycznej istotności różnic między jakością klasyfikatorów Wiemy, że możemy porównywad klasyfikatory np. za pomocą kroswalidacji.
Cystatin C as potential marker of Acute Kidney Injury in patients after Abdominal Aortic Aneurysms Surgery preliminary study
 Cystatin C as potential marker of Acute Kidney Injury in patients after Abdominal Aortic Aneurysms Surgery preliminary study Anna Bekier-Żelawska 1, Michał Kokot 1, Grzegorz Biolik 2, Damian Ziaja 2, Krzysztof
Cystatin C as potential marker of Acute Kidney Injury in patients after Abdominal Aortic Aneurysms Surgery preliminary study Anna Bekier-Żelawska 1, Michał Kokot 1, Grzegorz Biolik 2, Damian Ziaja 2, Krzysztof
Katowice, plan miasta: Skala 1: = City map = Stadtplan (Polish Edition)
 Katowice, plan miasta: Skala 1:20 000 = City map = Stadtplan (Polish Edition) Polskie Przedsiebiorstwo Wydawnictw Kartograficznych im. Eugeniusza Romera Click here if your download doesn"t start automatically
Katowice, plan miasta: Skala 1:20 000 = City map = Stadtplan (Polish Edition) Polskie Przedsiebiorstwo Wydawnictw Kartograficznych im. Eugeniusza Romera Click here if your download doesn"t start automatically
Network Services for Spatial Data in European Geo-Portals and their Compliance with ISO and OGC Standards
 INSPIRE Conference 2010 INSPIRE as a Framework for Cooperation Network Services for Spatial Data in European Geo-Portals and their Compliance with ISO and OGC Standards Elżbieta Bielecka Agnieszka Zwirowicz
INSPIRE Conference 2010 INSPIRE as a Framework for Cooperation Network Services for Spatial Data in European Geo-Portals and their Compliance with ISO and OGC Standards Elżbieta Bielecka Agnieszka Zwirowicz
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
Gdański Uniwersytet Medyczny Wydział Nauk o Zdrowiu z Oddziałem Pielęgniarstwa i Instytutem Medycyny Morskiej i Tropikalnej. Beata Wieczorek-Wójcik
 Gdański Uniwersytet Medyczny Wydział Nauk o Zdrowiu z Oddziałem Pielęgniarstwa i Instytutem Medycyny Morskiej i Tropikalnej Beata Wieczorek-Wójcik Poziom obsad pielęgniarskich a częstość i rodzaj zdarzeń
Gdański Uniwersytet Medyczny Wydział Nauk o Zdrowiu z Oddziałem Pielęgniarstwa i Instytutem Medycyny Morskiej i Tropikalnej Beata Wieczorek-Wójcik Poziom obsad pielęgniarskich a częstość i rodzaj zdarzeń
Has the heat wave frequency or intensity changed in Poland since 1950?
 Has the heat wave frequency or intensity changed in Poland since 1950? Joanna Wibig Department of Meteorology and Climatology, University of Lodz, Poland OUTLINE: Motivation Data Heat wave frequency measures
Has the heat wave frequency or intensity changed in Poland since 1950? Joanna Wibig Department of Meteorology and Climatology, University of Lodz, Poland OUTLINE: Motivation Data Heat wave frequency measures
Ocena wpływu nasilenia objawów zespołu nadpobudliwości psychoruchowej na masę ciała i BMI u dzieci i młodzieży
 Ewa Racicka-Pawlukiewicz Ocena wpływu nasilenia objawów zespołu nadpobudliwości psychoruchowej na masę ciała i BMI u dzieci i młodzieży Rozprawa na stopień doktora nauk medycznych PROMOTOR: Dr hab. n.
Ewa Racicka-Pawlukiewicz Ocena wpływu nasilenia objawów zespołu nadpobudliwości psychoruchowej na masę ciała i BMI u dzieci i młodzieży Rozprawa na stopień doktora nauk medycznych PROMOTOR: Dr hab. n.
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Fig 5 Spectrograms of the original signal (top) extracted shaft-related GAD components (middle) and
 Fig 4 Measured vibration signal (top). Blue original signal. Red component related to periodic excitation of resonances and noise. Green component related. Rotational speed profile used for experiment
Fig 4 Measured vibration signal (top). Blue original signal. Red component related to periodic excitation of resonances and noise. Green component related. Rotational speed profile used for experiment
Patients price acceptance SELECTED FINDINGS
 Patients price acceptance SELECTED FINDINGS October 2015 Summary With growing economy and Poles benefiting from this growth, perception of prices changes - this is also true for pharmaceuticals It may
Patients price acceptance SELECTED FINDINGS October 2015 Summary With growing economy and Poles benefiting from this growth, perception of prices changes - this is also true for pharmaceuticals It may
Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition)
 Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition) J Krupski Click here if your download doesn"t start automatically Karpacz, plan miasta 1:10 000: Panorama
Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition) J Krupski Click here if your download doesn"t start automatically Karpacz, plan miasta 1:10 000: Panorama
Analysis of Movie Profitability STAT 469 IN CLASS ANALYSIS #2
 Analysis of Movie Profitability STAT 469 IN CLASS ANALYSIS #2 aaaklnictzzjb9tgfmcnadpg7oy0lxa9edva9kkapdarhyk2k7gourinlwsweyzikuyiigvyleiv/cv767fpf/5crc1xt9va5mx7w3m/ecuqw1kuztpx/rl3/70h73/w4cog9dhhn3z62d6jzy+yzj766txpoir9nzszisjynetqr+rvlfvyoozu5xbybpsxb1wahul8phczdt2v4zgchb7uecwphlyigrgkjcyiflfyci0kxnmr4z6kw0jsokvot8isntpa3gbknlcufiv/h+hh+eur4fomd417rvtfjoit5pfju6yxiab2fmwk0y/feuybobqk+axnke8xzjjhfyd8kkpl9zdoddkazd5j6bzpemjb64smjb6vb4xmehysu08lsrszopxftlzee130jcb0zjxy7r5wa2f1s2off2+dyatrughnrtpkuprlcpu55zlxpss/yqe2eamjkcf0jye8w8yas0paf6t0t2i9stmcua+inbi2rt01tz22tubbqwidypvgz6piynkpobirkxgu54ibzoti4pkw2i5ow9lnuaoabhuxfxqhvnrj6w15tb3furnbm+scyxobjhr5pmj5j/w5ix9wsa2tlwx9alpshlunzjgnrwvqbpwzjl9wes+ptyn+ypy/jgskavtl8j0hz1djdhzwtpjbbvpr1zj7jpg6ve7zxfngj75zee0vmp9qm2uvgu/9zdofq6r+g8l4xctvo+v+xdrfr8oxiwutycu0qgyf8icuyvp/sixfi9zxe11vp6mrjjovpmxm6acrtbia+wjr9bevlgjwlz5xd3rfna9g06qytaoofk8olxbxc7xby2evqjmmk6pjvvzxmpbnct6+036xp5vdbrnbdqph8brlfn/n/khnfumhf6z1v7h/80yieukkd5j0un82t9mynxzmk0s/bzn4tacdziszdhwrl8x5ako8qp1n1zn0k6w2em0km9zj1i4yt1pt3xiprw85jmc2m1ut2geum6y6es2fwx6c+wlrpykblopbuj5nnr2byygfy5opllv4+jmm7s6u+tvhywbnb0kv2lt5th4xipmiij+y1toiyo7bo0d+vzvovjkp6aoejsubhj3qrp3fjd/m23pay8h218ibvx3nicofvd1xi86+kh6nb/b+hgsjp5+qwpurzlir15np66vmdehh6tyazdm1k/5ejtuvurgcqux6yc+qw/sbsaj7lkt4x9qmtp7euk6zbdedyuzu6ptsu2eeu3rxcz06uf6g8wyuveznhkbzynajbb7r7cbmla+jbtrst0ow2v6ntkwv8svnwqnu5pa3oxfeexf93739p93chq/fv+jr8r0d9brhpcxr2w88bvqbr41j6wvrb+u5dzjpvx+veoaxwptzp/8cen+xbg==
Analysis of Movie Profitability STAT 469 IN CLASS ANALYSIS #2 aaaklnictzzjb9tgfmcnadpg7oy0lxa9edva9kkapdarhyk2k7gourinlwsweyzikuyiigvyleiv/cv767fpf/5crc1xt9va5mx7w3m/ecuqw1kuztpx/rl3/70h73/w4cog9dhhn3z62d6jzy+yzj766txpoir9nzszisjynetqr+rvlfvyoozu5xbybpsxb1wahul8phczdt2v4zgchb7uecwphlyigrgkjcyiflfyci0kxnmr4z6kw0jsokvot8isntpa3gbknlcufiv/h+hh+eur4fomd417rvtfjoit5pfju6yxiab2fmwk0y/feuybobqk+axnke8xzjjhfyd8kkpl9zdoddkazd5j6bzpemjb64smjb6vb4xmehysu08lsrszopxftlzee130jcb0zjxy7r5wa2f1s2off2+dyatrughnrtpkuprlcpu55zlxpss/yqe2eamjkcf0jye8w8yas0paf6t0t2i9stmcua+inbi2rt01tz22tubbqwidypvgz6piynkpobirkxgu54ibzoti4pkw2i5ow9lnuaoabhuxfxqhvnrj6w15tb3furnbm+scyxobjhr5pmj5j/w5ix9wsa2tlwx9alpshlunzjgnrwvqbpwzjl9wes+ptyn+ypy/jgskavtl8j0hz1djdhzwtpjbbvpr1zj7jpg6ve7zxfngj75zee0vmp9qm2uvgu/9zdofq6r+g8l4xctvo+v+xdrfr8oxiwutycu0qgyf8icuyvp/sixfi9zxe11vp6mrjjovpmxm6acrtbia+wjr9bevlgjwlz5xd3rfna9g06qytaoofk8olxbxc7xby2evqjmmk6pjvvzxmpbnct6+036xp5vdbrnbdqph8brlfn/n/khnfumhf6z1v7h/80yieukkd5j0un82t9mynxzmk0s/bzn4tacdziszdhwrl8x5ako8qp1n1zn0k6w2em0km9zj1i4yt1pt3xiprw85jmc2m1ut2geum6y6es2fwx6c+wlrpykblopbuj5nnr2byygfy5opllv4+jmm7s6u+tvhywbnb0kv2lt5th4xipmiij+y1toiyo7bo0d+vzvovjkp6aoejsubhj3qrp3fjd/m23pay8h218ibvx3nicofvd1xi86+kh6nb/b+hgsjp5+qwpurzlir15np66vmdehh6tyazdm1k/5ejtuvurgcqux6yc+qw/sbsaj7lkt4x9qmtp7euk6zbdedyuzu6ptsu2eeu3rxcz06uf6g8wyuveznhkbzynajbb7r7cbmla+jbtrst0ow2v6ntkwv8svnwqnu5pa3oxfeexf93739p93chq/fv+jr8r0d9brhpcxr2w88bvqbr41j6wvrb+u5dzjpvx+veoaxwptzp/8cen+xbg==
HemoRec in Poland. Summary of bleeding episodes of haemophilia patients with inhibitor recorded in the years 2008 and 2009 04/2010
 HemoRec in Poland Summary of bleeding episodes of haemophilia patients with inhibitor recorded in the years 2008 and 2009 04/2010 Institute of Biostatistics and Analyses. Masaryk University. Brno Participating
HemoRec in Poland Summary of bleeding episodes of haemophilia patients with inhibitor recorded in the years 2008 and 2009 04/2010 Institute of Biostatistics and Analyses. Masaryk University. Brno Participating
European Crime Prevention Award (ECPA) Annex I - new version 2014
 European Crime Prevention Award (ECPA) Annex I - new version 2014 Załącznik nr 1 General information (Informacje ogólne) 1. Please specify your country. (Kraj pochodzenia:) 2. Is this your country s ECPA
European Crime Prevention Award (ECPA) Annex I - new version 2014 Załącznik nr 1 General information (Informacje ogólne) 1. Please specify your country. (Kraj pochodzenia:) 2. Is this your country s ECPA
Ocena potrzeb pacjentów z zaburzeniami psychicznymi
 Mikołaj Trizna Ocena potrzeb pacjentów z zaburzeniami psychicznymi przebywających na oddziałach psychiatrii sądowej Rozprawa na stopień doktora nauk medycznych Promotor: dr hab.n.med. Tomasz Adamowski,
Mikołaj Trizna Ocena potrzeb pacjentów z zaburzeniami psychicznymi przebywających na oddziałach psychiatrii sądowej Rozprawa na stopień doktora nauk medycznych Promotor: dr hab.n.med. Tomasz Adamowski,
DOI: / /32/37
 . 2015. 4 (32) 1:18 DOI: 10.17223/1998863 /32/37 -,,. - -. :,,,,., -, -.,.-.,.,.,. -., -,.,,., -, 70 80. (.,.,. ),, -,.,, -,, (1886 1980).,.,, (.,.,..), -, -,,,, ; -, - 346, -,.. :, -, -,,,,,.,,, -,,,
. 2015. 4 (32) 1:18 DOI: 10.17223/1998863 /32/37 -,,. - -. :,,,,., -, -.,.-.,.,.,. -., -,.,,., -, 70 80. (.,.,. ),, -,.,, -,, (1886 1980).,.,, (.,.,..), -, -,,,, ; -, - 346, -,.. :, -, -,,,,,.,,, -,,,
Lek. Ewelina Anna Dziedzic. Wpływ niedoboru witaminy D3 na stopień zaawansowania miażdżycy tętnic wieńcowych.
 Lek. Ewelina Anna Dziedzic Wpływ niedoboru witaminy D3 na stopień zaawansowania miażdżycy tętnic wieńcowych. Rozprawa na stopień naukowy doktora nauk medycznych Promotor: Prof. dr hab. n. med. Marek Dąbrowski
Lek. Ewelina Anna Dziedzic Wpływ niedoboru witaminy D3 na stopień zaawansowania miażdżycy tętnic wieńcowych. Rozprawa na stopień naukowy doktora nauk medycznych Promotor: Prof. dr hab. n. med. Marek Dąbrowski
Akademia Morska w Szczecinie. Wydział Mechaniczny
 Akademia Morska w Szczecinie Wydział Mechaniczny ROZPRAWA DOKTORSKA mgr inż. Marcin Kołodziejski Analiza metody obsługiwania zarządzanego niezawodnością pędników azymutalnych platformy pływającej Promotor:
Akademia Morska w Szczecinie Wydział Mechaniczny ROZPRAWA DOKTORSKA mgr inż. Marcin Kołodziejski Analiza metody obsługiwania zarządzanego niezawodnością pędników azymutalnych platformy pływającej Promotor:
Marta Uzdrowska. PRACA NA STOPIEŃ DOKTORA NAUK MEDYCZNYCH Promotor: Dr hab. n. med. prof. UM Anna Broniarczyk-Loba
 Marta Uzdrowska Ocena wad refrakcji i przyczyn niedowidzenia u osób z niepełnosprawnością intelektualną z wykorzystaniem zdalnego autorefraktometru Plusoptix PRACA NA STOPIEŃ DOKTORA NAUK MEDYCZNYCH Promotor:
Marta Uzdrowska Ocena wad refrakcji i przyczyn niedowidzenia u osób z niepełnosprawnością intelektualną z wykorzystaniem zdalnego autorefraktometru Plusoptix PRACA NA STOPIEŃ DOKTORA NAUK MEDYCZNYCH Promotor:
Formularz recenzji magazynu. Journal of Corporate Responsibility and Leadership Review Form
 Formularz recenzji magazynu Review Form Identyfikator magazynu/ Journal identification number: Tytuł artykułu/ Paper title: Recenzent/ Reviewer: (imię i nazwisko, stopień naukowy/name and surname, academic
Formularz recenzji magazynu Review Form Identyfikator magazynu/ Journal identification number: Tytuł artykułu/ Paper title: Recenzent/ Reviewer: (imię i nazwisko, stopień naukowy/name and surname, academic
Pielgrzymka do Ojczyzny: Przemowienia i homilie Ojca Swietego Jana Pawla II (Jan Pawel II-- pierwszy Polak na Stolicy Piotrowej) (Polish Edition)
 Pielgrzymka do Ojczyzny: Przemowienia i homilie Ojca Swietego Jana Pawla II (Jan Pawel II-- pierwszy Polak na Stolicy Piotrowej) (Polish Edition) Click here if your download doesn"t start automatically
Pielgrzymka do Ojczyzny: Przemowienia i homilie Ojca Swietego Jana Pawla II (Jan Pawel II-- pierwszy Polak na Stolicy Piotrowej) (Polish Edition) Click here if your download doesn"t start automatically
Porównanie skuteczności i bezpieczeństwa schematów immunoterapii BeG w raku pęcherza moczowego
 Lekarz Krzysztof Nowakowski Porównanie skuteczności i bezpieczeństwa schematów immunoterapii BeG w raku pęcherza moczowego Rozprawa na stopień doktora nauk medycznych Promotor: prof. dr hab. n. med. Andrzej
Lekarz Krzysztof Nowakowski Porównanie skuteczności i bezpieczeństwa schematów immunoterapii BeG w raku pęcherza moczowego Rozprawa na stopień doktora nauk medycznych Promotor: prof. dr hab. n. med. Andrzej
Zarządzanie sieciami telekomunikacyjnymi
 SNMP Protocol The Simple Network Management Protocol (SNMP) is an application layer protocol that facilitates the exchange of management information between network devices. It is part of the Transmission
SNMP Protocol The Simple Network Management Protocol (SNMP) is an application layer protocol that facilitates the exchange of management information between network devices. It is part of the Transmission
Hard-Margin Support Vector Machines
 Hard-Margin Support Vector Machines aaacaxicbzdlssnafiyn9vbjlepk3ay2gicupasvu4iblxuaw2hjmuwn7ddjjmxm1bkcg1/fjqsvt76fo9/gazqfvn8y+pjpozw5vx8zkpvtfxmlhcwl5zxyqrm2vrg5zw3vxmsoezi4ogkr6phieky5crvvjhriqvdom9l2xxftevuwcekj3lktmhghgniauiyutvrwxtvme34a77kbvg73gtygpjsrfati1+xc8c84bvraowbf+uwnipyehcvmkjrdx46vlykhkgykm3ujjdhcyzqkxy0chur6ax5cbg+1m4bbjptjcubuz4kuhvjoql93hkin5hxtav5x6yyqopnsyuneey5ni4keqrxbar5wqaxbik00icyo/iveiyqqvjo1u4fgzj/8f9x67bzmxnurjzmijtlybwfgcdjgfdtajwgcf2dwaj7ac3g1ho1n4814n7wwjgjmf/ys8fenfycuzq==
Hard-Margin Support Vector Machines aaacaxicbzdlssnafiyn9vbjlepk3ay2gicupasvu4iblxuaw2hjmuwn7ddjjmxm1bkcg1/fjqsvt76fo9/gazqfvn8y+pjpozw5vx8zkpvtfxmlhcwl5zxyqrm2vrg5zw3vxmsoezi4ogkr6phieky5crvvjhriqvdom9l2xxftevuwcekj3lktmhghgniauiyutvrwxtvme34a77kbvg73gtygpjsrfati1+xc8c84bvraowbf+uwnipyehcvmkjrdx46vlykhkgykm3ujjdhcyzqkxy0chur6ax5cbg+1m4bbjptjcubuz4kuhvjoql93hkin5hxtav5x6yyqopnsyuneey5ni4keqrxbar5wqaxbik00icyo/iveiyqqvjo1u4fgzj/8f9x67bzmxnurjzmijtlybwfgcdjgfdtajwgcf2dwaj7ac3g1ho1n4814n7wwjgjmf/ys8fenfycuzq==
Rozprawa na stopień naukowy doktora nauk medycznych w zakresie medycyny
 Lek. Maciej Jesionowski Efektywność stosowania budezonidu MMX u pacjentów z aktywną postacią łagodnego do umiarkowanego wrzodziejącego zapalenia jelita grubego w populacji polskiej. Rozprawa na stopień
Lek. Maciej Jesionowski Efektywność stosowania budezonidu MMX u pacjentów z aktywną postacią łagodnego do umiarkowanego wrzodziejącego zapalenia jelita grubego w populacji polskiej. Rozprawa na stopień
Rak trzustki cele terapeutyczne. Sekwencja leczenia.
 Rak trzustki cele terapeutyczne. Sekwencja leczenia. Leszek Kraj Klinika Hematologii, Onkologii i Chorób Wewnętrznych Warszawski Uniwersytet Medyczny Samodzielny Publiczny Centralny Szpital Kliniczny W
Rak trzustki cele terapeutyczne. Sekwencja leczenia. Leszek Kraj Klinika Hematologii, Onkologii i Chorób Wewnętrznych Warszawski Uniwersytet Medyczny Samodzielny Publiczny Centralny Szpital Kliniczny W
Machine Learning for Data Science (CS4786) Lecture11. Random Projections & Canonical Correlation Analysis
 Machine Learning for Data Science (CS4786) Lecture11 5 Random Projections & Canonical Correlation Analysis The Tall, THE FAT AND THE UGLY n X d The Tall, THE FAT AND THE UGLY d X > n X d n = n d d The
Machine Learning for Data Science (CS4786) Lecture11 5 Random Projections & Canonical Correlation Analysis The Tall, THE FAT AND THE UGLY n X d The Tall, THE FAT AND THE UGLY d X > n X d n = n d d The
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Stargard Szczecinski i okolice (Polish Edition)
 Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz Click here if your download doesn"t start automatically Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz
Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz Click here if your download doesn"t start automatically Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz
What our clients think about us? A summary od survey results
 What our clients think about us? A summary od survey results customer satisfaction survey We conducted our audit in June 2015 This is the first survey about customer satisfaction Why? To get customer feedback
What our clients think about us? A summary od survey results customer satisfaction survey We conducted our audit in June 2015 This is the first survey about customer satisfaction Why? To get customer feedback
Metodyki projektowania i modelowania systemów Cyganek & Kasperek & Rajda 2013 Katedra Elektroniki AGH
 Kierunek Elektronika i Telekomunikacja, Studia II stopnia Specjalność: Systemy wbudowane Metodyki projektowania i modelowania systemów Cyganek & Kasperek & Rajda 2013 Katedra Elektroniki AGH Zagadnienia
Kierunek Elektronika i Telekomunikacja, Studia II stopnia Specjalność: Systemy wbudowane Metodyki projektowania i modelowania systemów Cyganek & Kasperek & Rajda 2013 Katedra Elektroniki AGH Zagadnienia
Raport bieżący: 44/2018 Data: g. 21:03 Skrócona nazwa emitenta: SERINUS ENERGY plc
 Raport bieżący: 44/2018 Data: 2018-05-23 g. 21:03 Skrócona nazwa emitenta: SERINUS ENERGY plc Temat: Zawiadomienie o zmianie udziału w ogólnej liczbie głosów w Serinus Energy plc Podstawa prawna: Inne
Raport bieżący: 44/2018 Data: 2018-05-23 g. 21:03 Skrócona nazwa emitenta: SERINUS ENERGY plc Temat: Zawiadomienie o zmianie udziału w ogólnej liczbie głosów w Serinus Energy plc Podstawa prawna: Inne
Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2)
 Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Click here if your download doesn"t start automatically Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Emilka szuka swojej gwiazdy / Emily
Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Click here if your download doesn"t start automatically Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Emilka szuka swojej gwiazdy / Emily
No matter how much you have, it matters how much you need
 CSR STRATEGY KANCELARIA FINANSOWA TRITUM GROUP SP. Z O.O. No matter how much you have, it matters how much you need Kancelaria Finansowa Tritum Group Sp. z o.o. was established in 2007 we build trust among
CSR STRATEGY KANCELARIA FINANSOWA TRITUM GROUP SP. Z O.O. No matter how much you have, it matters how much you need Kancelaria Finansowa Tritum Group Sp. z o.o. was established in 2007 we build trust among
Health Resorts Pearls of Eastern Europe Innovative Cluster Health and Tourism
 Health Resorts Pearls of Eastern Europe Innovative Cluster Health and Tourism Projekt finansowany Fundusze Europejskie z budżetu państwa dla rozwoju oraz ze Polski środków Wschodniej Unii Europejskiej
Health Resorts Pearls of Eastern Europe Innovative Cluster Health and Tourism Projekt finansowany Fundusze Europejskie z budżetu państwa dla rozwoju oraz ze Polski środków Wschodniej Unii Europejskiej
Patient Protection in Clinical Trials
 Patient Protection in Clinical Trials Marek Czarkowski Bioethic Committee Warsaw Chamber of Physicians Factors affecting security and rights of research participants International regulations Domestic
Patient Protection in Clinical Trials Marek Czarkowski Bioethic Committee Warsaw Chamber of Physicians Factors affecting security and rights of research participants International regulations Domestic
Fizyka Procesów Klimatycznych Wykład 11 Aktualne zmiany klimatu: atmosfera, hydrosfera, kriosfera
 Fizyka Procesów Klimatycznych Wykład 11 Aktualne zmiany klimatu: atmosfera, hydrosfera, kriosfera prof. dr hab. Szymon Malinowski Instytut Geofizyki, Wydział Fizyki Uniwersytet Warszawski malina@igf.fuw.edu.pl
Fizyka Procesów Klimatycznych Wykład 11 Aktualne zmiany klimatu: atmosfera, hydrosfera, kriosfera prof. dr hab. Szymon Malinowski Instytut Geofizyki, Wydział Fizyki Uniwersytet Warszawski malina@igf.fuw.edu.pl
Revenue Maximization. Sept. 25, 2018
 Revenue Maximization Sept. 25, 2018 Goal So Far: Ideal Auctions Dominant-Strategy Incentive Compatible (DSIC) b i = v i is a dominant strategy u i 0 x is welfare-maximizing x and p run in polynomial time
Revenue Maximization Sept. 25, 2018 Goal So Far: Ideal Auctions Dominant-Strategy Incentive Compatible (DSIC) b i = v i is a dominant strategy u i 0 x is welfare-maximizing x and p run in polynomial time
Trend in drug use in Poland
 Prevalence and patterns of drug use among general population Indicator (GPS) Annual Expert Meeting 214 Trend in drug use in Poland Janusz Sierosławski Institute of Psychiatry i Neurology Warsaw Lisbon,
Prevalence and patterns of drug use among general population Indicator (GPS) Annual Expert Meeting 214 Trend in drug use in Poland Janusz Sierosławski Institute of Psychiatry i Neurology Warsaw Lisbon,
GDAŃSKI UNIWERSYTET MEDYCZNY
 GDAŃSKI UNIWERSYTET MEDYCZNY mgr Magdalena Pinkowicka WPŁYW TRENINGU EEG-BIOFEEDBACK NA POPRAWĘ WYBRANYCH FUNKCJI POZNAWCZYCH U DZIECI Z ADHD Rozprawa doktorska Promotor dr hab. n. med. Andrzej Frydrychowski,
GDAŃSKI UNIWERSYTET MEDYCZNY mgr Magdalena Pinkowicka WPŁYW TRENINGU EEG-BIOFEEDBACK NA POPRAWĘ WYBRANYCH FUNKCJI POZNAWCZYCH U DZIECI Z ADHD Rozprawa doktorska Promotor dr hab. n. med. Andrzej Frydrychowski,
CEE 111/211 Agenda Feb 17
 CEE 111/211 Agenda Feb 17 Tuesday: SW for project work: Jetstream, MSP, Revit, Riuska, POP, SV On R: drive; takes time to install Acoustics today: \\cife server\files\classes\cee111\presentations Thursday:
CEE 111/211 Agenda Feb 17 Tuesday: SW for project work: Jetstream, MSP, Revit, Riuska, POP, SV On R: drive; takes time to install Acoustics today: \\cife server\files\classes\cee111\presentations Thursday:
Spearman.
 2 1 * :... 109 - :. Spearman. (α=0/05). : 20-29.. 50. :.. : ). sheikhi@dnt.mui.ac.ir * ( :1. :2. 89/5/5 89/8/4. 89/11/1 573 568 :(5)6 :1389 568 : %75 : ) %35-40 :. %35 :. : ) (. ( ) ) ( ) -1 : ( -2 ( -3-4
2 1 * :... 109 - :. Spearman. (α=0/05). : 20-29.. 50. :.. : ). sheikhi@dnt.mui.ac.ir * ( :1. :2. 89/5/5 89/8/4. 89/11/1 573 568 :(5)6 :1389 568 : %75 : ) %35-40 :. %35 :. : ) (. ( ) ) ( ) -1 : ( -2 ( -3-4
Katarzyna Durda STRESZCZENIE STĘŻENIE KWASU FOLIOWEGO ORAZ ZMIANY W OBRĘBIE GENÓW REGULUJĄCYCH JEGO METABOLIZM JAKO CZYNNIK RYZYKA RAKA W POLSCE
 Pomorski Uniwersytet Medyczny Katarzyna Durda STRESZCZENIE STĘŻENIE KWASU FOLIOWEGO ORAZ ZMIANY W OBRĘBIE GENÓW REGULUJĄCYCH JEGO METABOLIZM JAKO CZYNNIK RYZYKA RAKA W POLSCE Promotor: dr hab. prof. nadzw.
Pomorski Uniwersytet Medyczny Katarzyna Durda STRESZCZENIE STĘŻENIE KWASU FOLIOWEGO ORAZ ZMIANY W OBRĘBIE GENÓW REGULUJĄCYCH JEGO METABOLIZM JAKO CZYNNIK RYZYKA RAKA W POLSCE Promotor: dr hab. prof. nadzw.
Towards Stability Analysis of Data Transport Mechanisms: a Fluid Model and an Application
 Towards Stability Analysis of Data Transport Mechanisms: a Fluid Model and an Application Gayane Vardoyan *, C. V. Hollot, Don Towsley* * College of Information and Computer Sciences, Department of Electrical
Towards Stability Analysis of Data Transport Mechanisms: a Fluid Model and an Application Gayane Vardoyan *, C. V. Hollot, Don Towsley* * College of Information and Computer Sciences, Department of Electrical
Linear Classification and Logistic Regression. Pascal Fua IC-CVLab
 Linear Classification and Logistic Regression Pascal Fua IC-CVLab 1 aaagcxicbdtdbtmwfafwdgxlhk8orha31ibqycvkdgpshdqxtwotng2pxtvqujmok1qlky5xllzrnobbediegwcap4votk2kqkf+/y/tnphdschtadu/giv3vtea99cfma8fpx7ytlxx7ckns4sylo3doom7jguhj1hxchmy/irhrlgh67lxb5x3blis8jjqynmedqujiu5zsqqagrx+yjcfpcrydusshmzeluzsg7tttiew5khhcuzm5rv0gn1unw6zl3gbzlpr3liwncyr6aaqinx4wnc/rpg6ix5szd86agoftuu0g/krjxdarph62enthdey3zn/+mi5zknou2ap+tclvhob9sxhwvhaqketnde7geqjp21zvjsfrcnkfhtejoz23vq97elxjlpbtmxpl6qxtl1sgfv1ptpy/yq9mgacrzkgje0hjj2rq7vtywnishnnkzsqekucnlblrarlh8x8szxolrrxkb8n6o4kmo/e7siisnozcfvsedlol60a/j8nmul/gby8mmssrfr2it8lkyxr9dirxxngzthtbaejv
Linear Classification and Logistic Regression Pascal Fua IC-CVLab 1 aaagcxicbdtdbtmwfafwdgxlhk8orha31ibqycvkdgpshdqxtwotng2pxtvqujmok1qlky5xllzrnobbediegwcap4votk2kqkf+/y/tnphdschtadu/giv3vtea99cfma8fpx7ytlxx7ckns4sylo3doom7jguhj1hxchmy/irhrlgh67lxb5x3blis8jjqynmedqujiu5zsqqagrx+yjcfpcrydusshmzeluzsg7tttiew5khhcuzm5rv0gn1unw6zl3gbzlpr3liwncyr6aaqinx4wnc/rpg6ix5szd86agoftuu0g/krjxdarph62enthdey3zn/+mi5zknou2ap+tclvhob9sxhwvhaqketnde7geqjp21zvjsfrcnkfhtejoz23vq97elxjlpbtmxpl6qxtl1sgfv1ptpy/yq9mgacrzkgje0hjj2rq7vtywnishnnkzsqekucnlblrarlh8x8szxolrrxkb8n6o4kmo/e7siisnozcfvsedlol60a/j8nmul/gby8mmssrfr2it8lkyxr9dirxxngzthtbaejv
Sargent Opens Sonairte Farmers' Market
 Sargent Opens Sonairte Farmers' Market 31 March, 2008 1V8VIZSV7EVKIRX8(1MRMWXIVSJ7XEXIEXXLI(ITEVXQIRXSJ%KVMGYPXYVI *MWLIVMIWERH*SSHTIVJSVQIHXLISJJMGMEPSTIRMRKSJXLI7SREMVXI*EVQIVW 1EVOIXMR0E]XS[R'S1IEXL
Sargent Opens Sonairte Farmers' Market 31 March, 2008 1V8VIZSV7EVKIRX8(1MRMWXIVSJ7XEXIEXXLI(ITEVXQIRXSJ%KVMGYPXYVI *MWLIVMIWERH*SSHTIVJSVQIHXLISJJMGMEPSTIRMRKSJXLI7SREMVXI*EVQIVW 1EVOIXMR0E]XS[R'S1IEXL
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
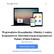 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Machine Learning for Data Science (CS4786) Lecture 11. Spectral Embedding + Clustering
 Machine Learning for Data Science (CS4786) Lecture 11 Spectral Embedding + Clustering MOTIVATING EXAMPLE What can you say from this network? MOTIVATING EXAMPLE How about now? THOUGHT EXPERIMENT For each
Machine Learning for Data Science (CS4786) Lecture 11 Spectral Embedding + Clustering MOTIVATING EXAMPLE What can you say from this network? MOTIVATING EXAMPLE How about now? THOUGHT EXPERIMENT For each
PROGRAM STAŻU. Nazwa podmiotu oferującego staż / Company name IBM Global Services Delivery Centre Sp z o.o.
 PROGRAM STAŻU Nazwa podmiotu oferującego staż / Company name IBM Global Services Delivery Centre Sp z o.o. Miejsce odbywania stażu / Legal address Muchoborska 8, 54-424 Wroclaw Stanowisko, obszar działania/
PROGRAM STAŻU Nazwa podmiotu oferującego staż / Company name IBM Global Services Delivery Centre Sp z o.o. Miejsce odbywania stażu / Legal address Muchoborska 8, 54-424 Wroclaw Stanowisko, obszar działania/
Wybrzeze Baltyku, mapa turystyczna 1: (Polish Edition)
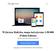 Wybrzeze Baltyku, mapa turystyczna 1:50 000 (Polish Edition) Click here if your download doesn"t start automatically Wybrzeze Baltyku, mapa turystyczna 1:50 000 (Polish Edition) Wybrzeze Baltyku, mapa
Wybrzeze Baltyku, mapa turystyczna 1:50 000 (Polish Edition) Click here if your download doesn"t start automatically Wybrzeze Baltyku, mapa turystyczna 1:50 000 (Polish Edition) Wybrzeze Baltyku, mapa
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
POLITECHNIKA WARSZAWSKA. Wydział Zarządzania ROZPRAWA DOKTORSKA. mgr Marcin Chrząścik
 POLITECHNIKA WARSZAWSKA Wydział Zarządzania ROZPRAWA DOKTORSKA mgr Marcin Chrząścik Model strategii promocji w zarządzaniu wizerunkiem regionu Warmii i Mazur Promotor dr hab. Jarosław S. Kardas, prof.
POLITECHNIKA WARSZAWSKA Wydział Zarządzania ROZPRAWA DOKTORSKA mgr Marcin Chrząścik Model strategii promocji w zarządzaniu wizerunkiem regionu Warmii i Mazur Promotor dr hab. Jarosław S. Kardas, prof.
Helena Boguta, klasa 8W, rok szkolny 2018/2019
 Poniższy zbiór zadań został wykonany w ramach projektu Mazowiecki program stypendialny dla uczniów szczególnie uzdolnionych - najlepsza inwestycja w człowieka w roku szkolnym 2018/2019. Składają się na
Poniższy zbiór zadań został wykonany w ramach projektu Mazowiecki program stypendialny dla uczniów szczególnie uzdolnionych - najlepsza inwestycja w człowieka w roku szkolnym 2018/2019. Składają się na
Ankiety Nowe funkcje! Pomoc magda.szewczyk@slo-wroc.pl. magda.szewczyk@slo-wroc.pl. Twoje konto Wyloguj. BIODIVERSITY OF RIVERS: Survey to students
 Ankiety Nowe funkcje! Pomoc magda.szewczyk@slo-wroc.pl Back Twoje konto Wyloguj magda.szewczyk@slo-wroc.pl BIODIVERSITY OF RIVERS: Survey to students Tworzenie ankiety Udostępnianie Analiza (55) Wyniki
Ankiety Nowe funkcje! Pomoc magda.szewczyk@slo-wroc.pl Back Twoje konto Wyloguj magda.szewczyk@slo-wroc.pl BIODIVERSITY OF RIVERS: Survey to students Tworzenie ankiety Udostępnianie Analiza (55) Wyniki
EDYTA KATARZYNA GŁAŻEWSKA METALOPROTEINAZY ORAZ ICH TKANKOWE INHIBITORY W OSOCZU OSÓB CHORYCH NA ŁUSZCZYCĘ LECZONYCH METODĄ FOTOTERAPII UVB.
 Wydział Farmaceutyczny z Oddziałem Medycyny Laboratoryjnej Uniwersytet Medyczny w Białymstoku EDYTA KATARZYNA GŁAŻEWSKA METALOPROTEINAZY ORAZ ICH TKANKOWE INHIBITORY W OSOCZU OSÓB CHORYCH NA ŁUSZCZYCĘ
Wydział Farmaceutyczny z Oddziałem Medycyny Laboratoryjnej Uniwersytet Medyczny w Białymstoku EDYTA KATARZYNA GŁAŻEWSKA METALOPROTEINAZY ORAZ ICH TKANKOWE INHIBITORY W OSOCZU OSÓB CHORYCH NA ŁUSZCZYCĘ
QUANTITATIVE AND QUALITATIVE CHARACTERISTICS OF FINGERPRINT BIOMETRIC TEMPLATES
 ZESZYTY NAUKOWE POLITECHNIKI ŚLĄSKIEJ 2014 Seria: ORGANIZACJA I ZARZĄDZANIE z. 74 Nr kol. 1921 Adrian KAPCZYŃSKI Politechnika Śląska Instytut Ekonomii i Informatyki QUANTITATIVE AND QUALITATIVE CHARACTERISTICS
ZESZYTY NAUKOWE POLITECHNIKI ŚLĄSKIEJ 2014 Seria: ORGANIZACJA I ZARZĄDZANIE z. 74 Nr kol. 1921 Adrian KAPCZYŃSKI Politechnika Śląska Instytut Ekonomii i Informatyki QUANTITATIVE AND QUALITATIVE CHARACTERISTICS
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Oznaczanie CA125 po I linii Pro i kontra. Krzysztof Sodowski vs Radosław Mądry Klinika Onkologii Uniwersytetu Medycznego w Poznaniu
 Oznaczanie CA125 po I linii Pro i kontra Krzysztof Sodowski vs Radosław Mądry Klinika Onkologii Uniwersytetu Medycznego w Poznaniu Early treatment of relapsed ovarian cancer based on CA125 level alone
Oznaczanie CA125 po I linii Pro i kontra Krzysztof Sodowski vs Radosław Mądry Klinika Onkologii Uniwersytetu Medycznego w Poznaniu Early treatment of relapsed ovarian cancer based on CA125 level alone
A DIFFERENT APPROACH WHERE YOU NEED TO NAVIGATE IN THE CURRENT STREAMS AND MOVEMENTS WHICH ARE EMBEDDED IN THE CULTURE AND THE SOCIETY
 A DIFFERENT APPROACH WHERE YOU NEED TO NAVIGATE IN THE CURRENT STREAMS AND MOVEMENTS WHICH ARE EMBEDDED IN THE CULTURE AND THE SOCIETY ODMIENNE PODEJŚCIE JAK NAWIGOWAĆ W OBECNYCH NURTACH I RUCHACH, KTÓRE
A DIFFERENT APPROACH WHERE YOU NEED TO NAVIGATE IN THE CURRENT STREAMS AND MOVEMENTS WHICH ARE EMBEDDED IN THE CULTURE AND THE SOCIETY ODMIENNE PODEJŚCIE JAK NAWIGOWAĆ W OBECNYCH NURTACH I RUCHACH, KTÓRE
Latent Dirichlet Allocation Models and their Evaluation IT for Practice 2016
 Latent Dirichlet Allocation Models and their Evaluation IT for Practice 2016 Paweł Lula Cracow University of Economics, Poland pawel.lula@uek.krakow.pl Latent Dirichlet Allocation (LDA) Documents Latent
Latent Dirichlet Allocation Models and their Evaluation IT for Practice 2016 Paweł Lula Cracow University of Economics, Poland pawel.lula@uek.krakow.pl Latent Dirichlet Allocation (LDA) Documents Latent
Poland) Wydawnictwo "Gea" (Warsaw. Click here if your download doesn"t start automatically
 Suwalski Park Krajobrazowy i okolice 1:50 000, mapa turystyczno-krajoznawcza =: Suwalki Landscape Park, tourist map = Suwalki Naturpark,... narodowe i krajobrazowe) (Polish Edition) Click here if your
Suwalski Park Krajobrazowy i okolice 1:50 000, mapa turystyczno-krajoznawcza =: Suwalki Landscape Park, tourist map = Suwalki Naturpark,... narodowe i krajobrazowe) (Polish Edition) Click here if your
Wczesny i zaawansowany rak piersi
 Warszawa, 14.12.2017 Wczesny i zaawansowany rak piersi Dr n. med. Agnieszka Jagiełło-Gruszfeld 1 Breast Cancer (C50): 1971-2011 Age-Standardised One-Year Net Survival, England and Wales Please include
Warszawa, 14.12.2017 Wczesny i zaawansowany rak piersi Dr n. med. Agnieszka Jagiełło-Gruszfeld 1 Breast Cancer (C50): 1971-2011 Age-Standardised One-Year Net Survival, England and Wales Please include
ZGŁOSZENIE WSPÓLNEGO POLSKO -. PROJEKTU NA LATA: APPLICATION FOR A JOINT POLISH -... PROJECT FOR THE YEARS:.
 ZGŁOSZENIE WSPÓLNEGO POLSKO -. PROJEKTU NA LATA: APPLICATION FOR A JOINT POLISH -... PROJECT FOR THE YEARS:. W RAMACH POROZUMIENIA O WSPÓŁPRACY NAUKOWEJ MIĘDZY POLSKĄ AKADEMIĄ NAUK I... UNDER THE AGREEMENT
ZGŁOSZENIE WSPÓLNEGO POLSKO -. PROJEKTU NA LATA: APPLICATION FOR A JOINT POLISH -... PROJECT FOR THE YEARS:. W RAMACH POROZUMIENIA O WSPÓŁPRACY NAUKOWEJ MIĘDZY POLSKĄ AKADEMIĄ NAUK I... UNDER THE AGREEMENT
Wydział Fizyki, Astronomii i Informatyki Stosowanej Uniwersytet Mikołaja Kopernika w Toruniu
 IONS-14 / OPTO Meeting For Young Researchers 2013 Khet Tournament On 3-6 July 2013 at the Faculty of Physics, Astronomy and Informatics of Nicolaus Copernicus University in Torun (Poland) there were two
IONS-14 / OPTO Meeting For Young Researchers 2013 Khet Tournament On 3-6 July 2013 at the Faculty of Physics, Astronomy and Informatics of Nicolaus Copernicus University in Torun (Poland) there were two
Maciej Pawlak. Zastosowanie nowoczesnych implantów i technik operacyjnych w leczeniu przepuklin brzusznych.
 Maciej Pawlak Zastosowanie nowoczesnych implantów i technik operacyjnych w leczeniu przepuklin brzusznych. Prospektywne randomizowane badanie porównujące zastosowanie dwóch różnych koncepcji siatki i staplera
Maciej Pawlak Zastosowanie nowoczesnych implantów i technik operacyjnych w leczeniu przepuklin brzusznych. Prospektywne randomizowane badanie porównujące zastosowanie dwóch różnych koncepcji siatki i staplera
Machine Learning for Data Science (CS4786) Lecture 24. Differential Privacy and Re-useable Holdout
 Machine Learning for Data Science (CS4786) Lecture 24 Differential Privacy and Re-useable Holdout Defining Privacy Defining Privacy Dataset + Defining Privacy Dataset + Learning Algorithm Distribution
Machine Learning for Data Science (CS4786) Lecture 24 Differential Privacy and Re-useable Holdout Defining Privacy Defining Privacy Dataset + Defining Privacy Dataset + Learning Algorithm Distribution
lek. Wojciech Mańkowski Kierownik Katedry: prof. zw. dr hab. n. med. Edward Wylęgała
 lek. Wojciech Mańkowski Zastosowanie wzrokowych potencjałów wywołanych (VEP) przy kwalifikacji pacjentów do zabiegu przeszczepu drążącego rogówki i operacji zaćmy Rozprawa na stopień doktora nauk medycznych
lek. Wojciech Mańkowski Zastosowanie wzrokowych potencjałów wywołanych (VEP) przy kwalifikacji pacjentów do zabiegu przeszczepu drążącego rogówki i operacji zaćmy Rozprawa na stopień doktora nauk medycznych
SubVersion. Piotr Mikulski. SubVersion. P. Mikulski. Co to jest subversion? Zalety SubVersion. Wady SubVersion. Inne różnice SubVersion i CVS
 Piotr Mikulski 2006 Subversion is a free/open-source version control system. That is, Subversion manages files and directories over time. A tree of files is placed into a central repository. The repository
Piotr Mikulski 2006 Subversion is a free/open-source version control system. That is, Subversion manages files and directories over time. A tree of files is placed into a central repository. The repository
Krytyczne czynniki sukcesu w zarządzaniu projektami
 Seweryn SPAŁEK Krytyczne czynniki sukcesu w zarządzaniu projektami MONOGRAFIA Wydawnictwo Politechniki Śląskiej Gliwice 2004 SPIS TREŚCI WPROWADZENIE 5 1. ZARZĄDZANIE PROJEKTAMI W ORGANIZACJI 13 1.1. Zarządzanie
Seweryn SPAŁEK Krytyczne czynniki sukcesu w zarządzaniu projektami MONOGRAFIA Wydawnictwo Politechniki Śląskiej Gliwice 2004 SPIS TREŚCI WPROWADZENIE 5 1. ZARZĄDZANIE PROJEKTAMI W ORGANIZACJI 13 1.1. Zarządzanie
