Received 25 March 2010; Accepted 19 October 2011
|
|
|
- Milena Wanda Kowal
- 8 lat temu
- Przeglądów:
Transkrypt
1 Journal of Apicultural Science 5 MICROSCOPIC IMAGE OF HONEYBEE DRONE SPERMATOZOA IN THREE DILUENTS G r z e g o r z B o r s u k 1, K r z y s z t o f O l s z e s k i 1, A n e t a S t r a c h e c k a 1, J e r z y P a l e o l o g 1, M a ri u s z G a g o 2, 3 1 Department of Biological Basis of Animal Production, Faculty of Biology and Animal Breeding, 2 Department of Biophysics, University of Life Sciences in Lublin, Akademicka 13, Lublin, Poland 3 Department of Cell Biology, Institute of Biology and Biotechnology, Maria Curie Sk odowska University, Lublin, Poland grzegorz.borsuk@up.lublin.pl INTRODUCTION Communities of social insects are characterised by a polyandric mating system (Strassmann, 2001). In natural mating, a queen bee may copulate with many drones (Ruttner, 1969; Moritz, et al., 1996; Schl ns, et al., 2004). This is related to polyandry (Triasko, 1951; Borsuk et al., 2011a,b), which is most highly organized in bees (Fuchs and Moritz, 1999; Koeniger and Koeniger, 2000; Palmer and Oldroyd, 2000). Natural mating occurs in the air, which means a person cannot control the Received 25 March 2010; Accepted 19 October 2011 S u m m a r y Microscopic analysis of the behaviour of spermatozoa diluted in diluents was conducted in vitro and in vivo. Additionally, an attempt was made to determine the factors that may affect semen coagulation in the instrumental insemination process. Under a microscope, the following semen smear groups were observed: 1. according to drone age; 2. in commercial diluents: saline, SAFE boar semen diluents; l semen with spermathecal uid from 4-6 virgin queen bees; 4. from partially lled spermatheca - we used spermatozoa after reinsemination. Twelve smears in each group were viewed under the OLYMPUS microscope. The microscope was coupled with a digital camera for lming and taking photographs of the semen. Motility of spermatozoa/semen was observed in the preparations. A stopwatch was used to measure the length of time between preparations and the beginning of sperm movement until full cessation of movement. Semen from young drones did not coagulate. Semen coagulation most frequently occurred (44%) when mixing the semen from old and mature drones. After 12 min, cessation of spermatozoa movement was observed in the semen diluted with saline. Spermatozoa in the boar semen diluents exhibited motility for 29 min. After reinsemination, spermatozoa displayed circular arrangement. Key ords: semen smears, semen coagulation, reinsemination, drone, Apis mellifera. choice of the paternal side. Improved methods of instrumental bee insemination, however, have provided full control over the selection of the paternal side during mating. This significantly contributed to progress in bee breeding. Still, it is not known why instrumentally inseminated queen bees do not always commence oviposition. It is also not known why queens who have been instrumentally inseminated, start egg-laying later than naturallyinseminated queens (Mackensen, 1951, 1964; Jasi ski et al., 2005; Woyke et al., 2008).
2 6 In the past, the efficiency of properly performed instrumental insemination was affected by semen quality and lack of drone-specific diluents as well as other factors. Nowadays though, their analogues are applied with a common 0.9% saline solution and boar semen diluents. However, most diluents or substances are exposed to semen, reducing the survival rate of spermatozoa (Bishop, 1920). This explains why an air space in the insemination needle is necessary during the instrumental insemination. It prevents semen contact with the liquid in the needle and syringe. Another factor that affects the efficiency of the properly performed insemination procedure is semen competition. Inseminators claim that semen tends to coagulate in the needle during numerous insemination processes (Loc personal information). This coagulation markedly prolongs the insemination process. Internal complications may occur after natural mating between the queen bee and several drones (Schl ns et al., 2004; Andere et al., 2011). The internal complications may be caused by one drone, which, for instance, is the first to mate and contributes to semen coagulation. Semen coagulation consequently affects further sperm storage (Pizzari and Foster, 2008). After the mating, spermatozoa may compete between ejaculates from different drones (polyandry), which may induce their coagulation. The coagulation may be caused by incompatibility in the sex locus (Mackensen, 1964) as well as population selection. If this is so, none of the semen can be used since it remains in the queen bee s reproductive tract (Wojciechowski and Kr l, 1996). The survival of the semen stored by the queen bee is affected by the spermathecal fluid (Verma, 1973, 1978) and by substances secreted by the spermathecal glands (Schl ns et al., 2004). The spermatheca contains antioxidative enzymes protecting the spermatozoa from oxidative stress (Weirich et al., 2002). It is possible that such stress is induced by competition between the spermatozoa (Triasko, 1951). The competition involves increased motility, ensuring sperm precedence in reaching the queen bee s spermatheca. Another reason for lack of oviposition may be aggregation of spermatozoa from one drone, which form unbreakable bundles (Moritz et al., 1996). After copulation, the continually mixing spermatozoa are well visible through the transparent spermathecal wall (Ruttner, 1954; Woyke, 1960). After insemination with mixed semen originating from African and European drones, a higher rate of egg fertilization by African drones was observed. The ratio was 9:1 (Grandi-Hoffman et al., 2004; Schneider et al., 2006). Similar dominance was noted in wild drones and in those selected in the drone congregation areas with the progeny ratio 4:1, respectively (Woyke, 1971). This indicates high competition between spermatozoa (Tofilski et al., 2011). The introduction sequence of semen into a queen bee s reproductive tract also plays an important role. There is little probability of progeny from drones that were the last to be used for insemination (Woyke, 1963; Siuda et al., 2009). This is because excess semen from the queen bee s reproductive tract is removed. The techniques employed in the evaluation of the semen include the macroscopic method and the in vivo microscopic method. The macroscopic method uses the consistency estimation according to Woyke (1964). Woyke classifies semen into: 1) sparse semen, which is not suitable for insemination, 2) semen that is optimal for insemination, and 3) dense semen - also not suitable for insemination. Semen used in instrumental bee insemination can be assessed with the in vivo microscopic method based on the number of laid eggs (Jasi ski et al., 2005; Woyke et al., 2008). The aim of the experiment was to perform an in vitro and in vivo microscopic
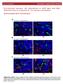
3 Journal of Apicultural Science 7 analysis of diluted spermatozoa behaviour in diluents. Additionally, an attempt was made to determine the factors that may affect semen coagulation in the instrumental insemination process. MATERIALS AND METHODS The study was conducted in the Breeding Apiary in Teodor w, Poland. Carniolan honeybee Apis mellifera carnica drones and queen bees were investigated. The following semen smear groups were observed under the microscope: 1. according to drone age; Three age groups were distinguished: young drones up to 12 days old (sparse semen), mature drones which were from 14 to 27 days old (optimal semen for instrumental insemination), and older drones which were over 30 days old (dense semen); 2. in commercial diluents: saline, SAFE boar semen diluents; l semen with the spermathecal fluid from 4-6 virgin queen bees; 4. from partially filled spermatheca. The queen bees were inseminated by one drone with a dose of approximately 1 l. After commencement of oviposition the semen was collected from the spermatheca. Then, this semen was used for reinsemination of twelve virgin queens using a dose of approximately 0.5 l. The semen smears from the partially filled spermatheca and the spermatozoa after the reinsemination, were prepared to assess the behaviour of the spermatozoa in the spermatheca. The semen was mixed with diluents in a ratio of about 1:4. The smears were prepared on a B rker s plate. Twelve smears were made from each group. The smears were observed under an OLYMPUS microscope. The microscope was coupled with a digital camera to film the observed semen. Motility of spermatozoa/semen was observed in the preparations. A stopwatch was used to measure the length between the time the preparation was made and the beginning of spermatozoa/ semen movement - until full cessation of movement. The mean time of spermatozoa/semen movement in the preparations was calculated with SAS software (Institute, SAS User s Guide Version 9.1 ed., 2003) using univariate ANOVA. RESULTS AND DISCUSSION 1. Behaviour of the semen in relation to drone age Sparse semen from young drones had a cluster visible in loose spermatozoa bundles and short mobility duration (X SD= min, n=12) (Fig. 1). Similarly, Jaycox (1960), Lensky and Schindler (1967), Ruttner (1969) and Verma (1978) assessed the spermatozoa after cessation of movement. The optimal semen for instrumental insemination Fig. 1. Semen from a young drone.
4 8 Fig. 2. Semen from a mature drone. Fig. 3. Semen from an old drone. exhibited long mobility duration (X SD= min, n=12) and was accompanied by a high cluster mass of spermatozoa bonds (Fig. 2). Dense semen sampled from old drones displayed short mobility duration (X SD= min, n=12) in the microscopic picture (Fig. 3). This dense semen had a very high cluster mass of spermatozoa and varied mobility duration. Dense semen demonstrated high diversity and had a tendency to coagulate. The tendency to coagulate lowered the motility of the spermatozoa and induced their immobility. In the non-coagulated semen, movements of the spermatozoa were still visible. The mixed semen from two mature drones was very active but this activity later disappeared. Such reactions may indicate competition between the ejaculates (Triasko, 1951). Similar behaviour was observed in a mixture of ejaculates sampled from a young and a mature drone. At first, enhanced motility was observed. This enhanced motility may have been induced by an increased amount of the seminal vesicle fluid and of the endophallus bulb (Flanders, 1939; Woyke, 2008) or proteins (King et al., 2011). Sparse semen from immature drones did not coagulate when combined with the optimal consistency semen used
5 Journal of Apicultural Science 9 for insemination. The lack of semen coagulation may indicate that there is no hemolymph and endothelium in the young drone semen (Ruttner, 1954; Woyke, 2008, 2010). Dense semen caused rapid loss of motility and more frequent (44%) production of coagulates, not motility bands. The cessation of semen movement may have been induced by the increased amount of epithelium entering the semen (Woyke, 2008, 2010). Such an increase in epithelium may be a result of the drone aging process (Burzy ski, 2007; Rhodes et al., 2010; Paleolog et al., 2011). It may also result from the peeling of considerable amounts of the epithelium from mucus glands during eversion of the endophallus. 2. Behaviour of the semen in commercial diluents The semen from the young, mature and old drones was treated with a salinedilution. During the treatment, the spermatozoa displayed quick movements, which on average, ceased after X SD= min, n=12. Movement cessation was an indication of dying spermatozoa (Ruttner, 1969). Dilution resulted in disruption of the bonds and the first characteristic circular arrangement. Fig. 4. Semen from a mature drone mixed with the spermathecal uid of a non-inseminated queen bee (0,2 l semen with the spermathecal uid from 4-6 virgin queen bees). Fig. 5. Spermatozoa after reinsemination.
6 10 The boar semen diluent also induced increased spermatozoa motility accompanied by almost complete disruption of the bonds of the spermatozoa, which then restored the bonds. On average, during X SD= min, n=12 period, single spermatozoa at the sides of the preparations displayed the highest motility. 3. Behaviour of the semen in combination ith the spermathecal fluid Semen sampled from a mature drone and mixed with the spermathecal liquid of a non-inseminated queen bee initially displayed intensive motility. The mobility declined after a few minutes and remained at a constant level for average X SD= min, n=12 (Fig. 4). After the queen bees were inseminated with such a mixture and smears from their spermatheca were prepared, it appeared that the spermatozoa were arranged in loose bundles. These loose bundles formed circles which moved in a counter clockwise manner (Fig. 4). King et al. (2011) argue that sperm viability is dependent on protein and fructose found in seminal fluid whereas Gen er and Kahya (2011) state that sperm viability is dependent on lateral oviducts and spermathecae substances. 4. Behaviour of the semen sampled from the partially filled spermatheca In the partially filled spermatheca, highly motile spermatozoa arranged in the form of loose clusters were observed. Such spermatozoa have the ability of unconstrained movement and are not arranged in a cluster mass of spermatozoa bonds (Ruttner, 1954; Woyke, 2008, 2010), therefore their activity is increased. The higher the cluster of the spermatozoa in the spermatheca is, the more compact the bundles are. This means that the spermatozoa remain in tight clusters without being able to move. Characteristic behaviour was observed in the spermatozoa sampled from the spermatheca of the queen bees after reinsemination. The spermathecae were partially filled due to the small amount of semen sampled from a previously inseminated ovipositing queen. In all the smears, the spermatozoa formed more or less compact circles. The compactness depended on the amount of semen sampled from the spermatheca of an ovipositing queen bee (ca. 0.5 l). All the spermatozoa were arranged in circles, which displayed a rotary counter clockwise motion (all moving in the same direction) (Fig. 5). The circle arrangement may be related to the length of the spermatozoa, which is ca. mm (Ruttner, 1954). The circle arrangement facilitates storage of the long, circularly arranged spermatozoa in small, round, 1 mm in diameter spermathecae. The arrangement of the spermatozoa may also be related to polyandry. When sampled from several drones, the spermatozoa form circular clusters, mix in the queen bee s spermatheca (Ruttner, 1954; Woyke, 1960), and move into the vas deferens. Thus, the circular clusters close the vas deference temporarily until they become scarce. Other circular clusters then take their place. This is corroborated by the fact, that fertilized eggs laid by a queen bee recurrently descend from a different drone. A very visible example is in a bee family with naturally mated dark-coloured bees, where occasionally light-coloured bees appear after the queen bee has copulated with several light-coloured drones. CONCLUSION Semen from mature drones coagulates the earliest. Semen from young drones does not coagulate. ACKNOwLEDGEMENTS We thank Prof. Jerzy Woyke for critical support and constructive comments during the preparation of the manuscript. We also wish to thank Mr. Krzysztof Loc for his invaluable help in conducting the experiments.
7 Journal of Apicultural Science 11 REFFERENCES Andere C. I., Monteavaro C., Palacio M. A., Catena M., Rodr guez E. M., Collins A. M. (2011) - Apis mellifera semen: bacterial contamination and susceptibility to antibiotics. Apidologie, 42(5): Bishop G. H. (1920) - Fertilization in the honeybee. I. The male sexual organs: their histological structure and physiological functioning. J. Exp. Zool., 31: Borsuk G., Olszewski K., Strachecka A., Paleolog J. (2011a) - The interaction of worker bees with elevated genotype variance 1. Field tests of sugar syrup collection and storage. J. Apic. Sci., 55(1): Borsuk G., Olszewski K., Strachecka A., Paleolog J. (2011b) - The interaction of worker bees with elevated genotype variance 2. Cage tests of sugar syrup collecting and mortality. J. Apic. Sci., 55(1): Burzy ski S. R. (2007) - Prze om w terapii i profilaktyce w medycynie XXI wieku (III). Medycyna starzej cego si spo ecze stwa [A breakthrough in therapy and prophylaxis in medicine twenty-first century (III). Aging Medicine]. Czasopismo Aptekarskie, 1: Flanders S. E. (1939) - Environmental control of sex in Hymenopterous insects. Ann. Ent. Soc. Amer., 32(1): Fuchs S., Moritz R. F. A. (1999) - Evolution of extreme polyandry in the honeybee Apis mellifera. Behav. Ecol. Sociobiol., 45(3-4): Gen er H. V., Kahya Y. (2011) - The viability of sperm in lateral oviducts and spermathecae of instrumentally inseminated and naturally mated honey bee (Apis mellifera L.) queens. J. Apic. Res., 50(3): Grandi-Hoffman de G., Tarpy D. R., Schneider S. S. (2004) - Details, details: How sperm use might influence the Africanization process. Am. Bee J., 144(6): Jasi ski J., Prabucki J., Wilde J., Woyke J., Chuda-Mickiewicz B., Siuda M., Madras B., Samborski J., Bratkowski J., Jojczyk A., B k B. (2005) - Badania nad czynnikami przy pieszaj cymi czerwienie sztucznie unasienionych matek pszczelich II [Studies on factors accelerating oviposition of instrumentally inseminated queen bees II]. XLII Naukowa Konf. Pszczelarska, Pu awy, Jaycox E. R. (1960) - The effect of drying and various diluents on spermatozoa of honey bee (Apis mellifera L.). J. Econ. Entomol., 53(2): King M., Eubel H., Millar A. H., Baer B. (2011) - Proteins within the seminal fluid are crucial to keep sperm viable in the honeybee Apis mellifera. J. Insect. Physiol., 57(3): Koeniger N., Koeniger G. (2000) - Reproductive isolation among species of the genus Apis. Apidologie, 31(2): Lensky Y., Schindler H. (1967) - Motility and reversible inactivation of honeybee spermatozoa in vivo and in vitro. Ann. Abeille, 10(1): Mackensen O. (1951) - Viability and sex determination in the honeybee. Genetics, 36(5): Mackensen O. (1964) - Relation of semen volume to success in artificial insemination. J. Econ. Entomol., 57(4): Moritz R. F. A, Kryger P., Allsopp M. H. (1996) - Competition for royalty in bees. Nature, 384: 31. Moritz R. F. A. (1983) - Homogeneous mixing of honeybee semen by centrifugation. J. Apic. Res., 22(4): Paleolog J., Strachecka A., Burzy ski S. R., Olszewski K., Borsuk G. (2011) - The larval diet supplemented with sodium phenylacetylglutaminate influences the worker cuticle proteolytic system in Apis mellifera L. J. Apic. Sci., 55(2): Palmer K. A., Oldroyd B. P. (2000) - Evolution of multiple mating in the genus Apis. Apidologie, 31(2):
8 12 Pizzari T., Foster K. R. (2008) - Sperm sociality: Cooperation, altruizm, and spite. PLoS Biol. 6 (5): e 130. cloi; /journal. pbio Rhodes J.W., Harden S., Spooner- Hart R., Anderson D. L., Wheen G. (2010) - Effects of age, season and genetics on semen and sperm production in Apis mellifera drones. Apidologie, 42(1): Ruttner F. (1954) - Mehrfache Begattung der Bienenk nigin. Zool. Anz., 153: Ruttner F. (1969) - The instrumental insemination of the queen bees. Apimondia Verlag, Bukarest, Romania. SAS Institute (2003) - SAS; Statistic s. SAS Institute Inc., Cary, NC, USA. Schl ns H., Koeniger G., Koeniger N., Moritz R. F. A. (2004) - Sperm utilization pattern in the honeybee (Apis mellifera). Behav. Ecol. Sociobiol., 56: Schneider S. S., de Grandi-Hoffman G., Smith D., Tarpy D. (2006) - The African honey bee. I. A case study of a biological invasion. Bee Culture, 134(4): Siuda M., B k B., Wilde J. (2009) - The impact of the sequence of particular drone semen administration on number of their own progeny after instrumental insemination Ann. Warsaw Univ. of Life Sc. - SGGW, Anim. Sci., 46: Strassmann J. E. (2001) - The rarity of multiple mating by females in the social hymenoptera. Insectes Sociaux, 48(1): Tofilski A., Chuda-Mickiewicz B., Czeko ska K., Chorobi ski P. (2011) - Flow cytometry evidence about sperm competition in honey bee (Apis mellifera). Apidologie, DOI: /s Triasko W. W. (1951) - Priznaki osiemiennosti pczelinych matok [Signs ofmating of queen bees]. Pchelovodstvo, 28(11): Verma L. R. (1973) - An ionic basis for a possible mechanism of sperm survival in the spermatheca of the queen honey bee (Apis mellifera L.). Comp. Biochem. and Physiol., 44(1): Verma L. R. (1978) - Biology of honeybee (Apis mellifera L.) spermatozoa 1. Effect of different diluents on motility and survival. Apidologie, 9(3): Weirich G., Collins, A., Williams V. (2002) - Antioxidant enzymes in the honey bee, Apis mellifera. Apidologie, 33(1): Wojciechowski M., Kr l E. (1996) - On intraoviductal sperm competition in the honeybee (Apis mellifera). Folia Biol. Krakow, 44: Woyke J. (1960) - Naturalne i sztuczne unasienianie matek pszczelich (Natural and artificial insemination of queen honey bees). Pszczeln. Zesz. Nauk., 4: Woyke J. (1963) - Contribution of successive drones to the insemination of a queen. XIX-th Intern. Beekeeping Congress, Prague. Proceedings, Comp. Text. of Lect. of XIX Congress of Apimondia: [online]: contrsuccdr.pdf Woyke J. (1964) - Die Virkung aufeinanderfolgender Drohnen auf die Besamung der K nigin. Beekeep. Congr. Prague, 19: Woyke J. (1971) - Unasienianie matek pszczelich na trutowisku o zwi kszonej liczbie trutni (The matings of honey bee queens in the mating station with enlarged drones numer). Pszczeln. Zesz. Nauk., 15(1-2): Woyke J. (2008) - Why the eversion of the endophallus of honey bee drone stops at the partly everted stage and significance of this. Apidologie, 39(6): Woyke J., Jasi ski Z., Prabucki J., Wilde J., Chuda-Mickiewicz B., Siuda M., Madras-Majewska B., Samborski J., Bratkowski J, Jojczyk A. (2008) - Onset of oviposition by honey bee queens, mated either naturally or by various instrumental insemination methods, fits a lognormal distribution. J. Apic. Res. and Bee World, 47(1): 1-9. Woyke J. (2010) - Three substances ejected by Apis mellifera drones during endophallus eversion as well as during natural matings with queen bees. Apidologie, 41(6):
9 Journal of Apicultural Science 13 MIKROSKOPOwY OBRAZ PLEMNIK w TRUTNI PSZCZO Y MIODNEJ w TRZECH ROZCIE CZALNIKACH B o r s u k G., O l s z e s k i K., S t r a c h e ck a A., P a l e o l o g J., G a g o M. S t r e s z c z e n i e Celem bada by a mikroskopowa analiza zachowania si plemnik w w rozcie czalnikach in vitro i in vivo. Dodatkowo pr bowano okre li, kt re z czynnik w mog wp ywa na koagulacj nasienia podczas instrumentalnego unasieniania. Badano trutnie i matki rasy krai skiej Apis mellifera carnica. Utworzono cztery grupy do wiadczalne. Nasienie obserwowano w rozmazach mikroskopowych: 1. pobrane od trutni w r nym wieku. Wyodr bniono trzy grupy wiekowe: trutnie m ode - do 12-go dnia ycia (daj ce rzadkie nasienie), trutnie dojrza e - w przedziale wiekowym dni (daj ce nasienie optymalne do instrumentalnego unasieniania), trutnie stare - powy ej 30-stu dni (daj ce g ste nasienie). 2. z handlowymi rozcie czalnikami (sol zjologiczn oraz z rozcie czalnikiem nasienia knur w SAFE), 3. z p ynem pochodz cym ze zbiorniczka nasiennego dziewiczych matek, 4. ze zbiorniczka nasiennego matki, kt r unasieniano jednym trutniem (dawka ok. 1 l) i czekano, a zacznie sk ada jaja nast pnie matk zabijano i pobierano nasienie ze zbiorniczka nasiennego, kt re wykorzystywano do powt rnego unasieniania dziewiczej matki - reinseminacja (dawka ok. 0,5 l). Rozmaz plemnik w ze zbiorniczka nasiennego matki, po reinseminacji u yto w celu stwierdzenia zachowania si plemnik w w zbiorniczku nasiennym innej dziewiczej matki. Nasienie mieszano z rozcie czalnikami w proporcji 1:4. Rozmazy wykonywano na p ytce B rkera. W ka dej grupie wykonano po 12 rozmaz w, kt re ogl dano w mikroskopie OLYMPUS. Mikroskop po czono z kamer cyfrow, kt r nagrywano lmy nasienia i wykonano zdj cia. W preparatach obserwowano ruch plemnik w/nasienia. Stoperem mierzono czas od wykonania preparatu, pocz tek ruchu plemnik w/nasienia w preparacie do ca kowitego zaprzestania ruchu. Nasienie od m odych trutni nie powodowa o koagulat w. Najcz ciej do koagulacji nasienia (44%) dochodzi o po zmieszaniu nasienia pochodz cego od starych trutni z nasieniem od dojrza ych trutni. Po 12 min obserwowano zaprzestanie ruchu plemnik w w nasieniu rozcie czonym sol zjologiczn. Plemniki w rozcie czalniku dla knur w wykazywa y oznaki ruchu przez 29 min. Plemniki po reinseminacji uk ada y si w ko a. S o a kluczo e: rozmaz nasienia, koagulacja nasienia, reinseminacja, trutnie, Apis mellifera.
The impact of the sequence of particular drone semen administration on number of their own progeny after instrumental insemination
 Annals of Warsaw University of Life Sciences SGGW Animal Science No 46, 2009: 109 113 (Ann. Warsaw Univ. of Life Sc. SGGW, Anim. Sci. 46, 2009) The impact of the sequence of particular drone semen administration
Annals of Warsaw University of Life Sciences SGGW Animal Science No 46, 2009: 109 113 (Ann. Warsaw Univ. of Life Sc. SGGW, Anim. Sci. 46, 2009) The impact of the sequence of particular drone semen administration
The influence of honey bee (Apis mellifera) drone age on volume of semen and viability of spermatozoa
 DOI: 10.2478/jas-2013-0007 Vol. 57 No. 1 2013 Journal of Apicultural Science 61 The influence of honey bee (Apis mellifera) drone age on volume of semen and viability of spermatozoa K r y s t y n a C z
DOI: 10.2478/jas-2013-0007 Vol. 57 No. 1 2013 Journal of Apicultural Science 61 The influence of honey bee (Apis mellifera) drone age on volume of semen and viability of spermatozoa K r y s t y n a C z
Tychy, plan miasta: Skala 1: (Polish Edition)
 Tychy, plan miasta: Skala 1:20 000 (Polish Edition) Poland) Przedsiebiorstwo Geodezyjno-Kartograficzne (Katowice Click here if your download doesn"t start automatically Tychy, plan miasta: Skala 1:20 000
Tychy, plan miasta: Skala 1:20 000 (Polish Edition) Poland) Przedsiebiorstwo Geodezyjno-Kartograficzne (Katowice Click here if your download doesn"t start automatically Tychy, plan miasta: Skala 1:20 000
SSW1.1, HFW Fry #20, Zeno #25 Benchmark: Qtr.1. Fry #65, Zeno #67. like
 SSW1.1, HFW Fry #20, Zeno #25 Benchmark: Qtr.1 I SSW1.1, HFW Fry #65, Zeno #67 Benchmark: Qtr.1 like SSW1.2, HFW Fry #47, Zeno #59 Benchmark: Qtr.1 do SSW1.2, HFW Fry #5, Zeno #4 Benchmark: Qtr.1 to SSW1.2,
SSW1.1, HFW Fry #20, Zeno #25 Benchmark: Qtr.1 I SSW1.1, HFW Fry #65, Zeno #67 Benchmark: Qtr.1 like SSW1.2, HFW Fry #47, Zeno #59 Benchmark: Qtr.1 do SSW1.2, HFW Fry #5, Zeno #4 Benchmark: Qtr.1 to SSW1.2,
THE ROLE OF PHYTOHORMONES IN INSTRUMENTAL INSEMINATION OF QUEEN BEES
 Vol. 53 No. 2 2009 Journal of Apicultural Science 91 THE ROLE OF PHYTOHORMONES IN INSTRUMENTAL INSEMINATION OF QUEEN BEES B o e n a C h u d a - M i c k i e w i c z, J a r o s ³ a w P r a b u c k i, J e
Vol. 53 No. 2 2009 Journal of Apicultural Science 91 THE ROLE OF PHYTOHORMONES IN INSTRUMENTAL INSEMINATION OF QUEEN BEES B o e n a C h u d a - M i c k i e w i c z, J a r o s ³ a w P r a b u c k i, J e
Weronika Mysliwiec, klasa 8W, rok szkolny 2018/2019
 Poniższy zbiór zadań został wykonany w ramach projektu Mazowiecki program stypendialny dla uczniów szczególnie uzdolnionych - najlepsza inwestycja w człowieka w roku szkolnym 2018/2019. Tresci zadań rozwiązanych
Poniższy zbiór zadań został wykonany w ramach projektu Mazowiecki program stypendialny dla uczniów szczególnie uzdolnionych - najlepsza inwestycja w człowieka w roku szkolnym 2018/2019. Tresci zadań rozwiązanych
Helena Boguta, klasa 8W, rok szkolny 2018/2019
 Poniższy zbiór zadań został wykonany w ramach projektu Mazowiecki program stypendialny dla uczniów szczególnie uzdolnionych - najlepsza inwestycja w człowieka w roku szkolnym 2018/2019. Składają się na
Poniższy zbiór zadań został wykonany w ramach projektu Mazowiecki program stypendialny dla uczniów szczególnie uzdolnionych - najlepsza inwestycja w człowieka w roku szkolnym 2018/2019. Składają się na
OPRACOWANIE METODY KRÓTKOTERMINOWEGO PRZECHOWYWANIA NASIENIA TRUTNI. Wojciech Skowronek, Jerzy Szymula
 PSZCZELNICZE ZESZYTY NAUKOWE Rok XLII, Nr l 1998 OPRACOWANIE METODY KRÓTKOTERMINOWEGO PRZECHOWYWANIA NASIENIA TRUTNI Wojciech Skowronek, Jerzy Szymula Instytut Sadownictwa i Kwiaciarstwa, Oddział Pszczelnictwa
PSZCZELNICZE ZESZYTY NAUKOWE Rok XLII, Nr l 1998 OPRACOWANIE METODY KRÓTKOTERMINOWEGO PRZECHOWYWANIA NASIENIA TRUTNI Wojciech Skowronek, Jerzy Szymula Instytut Sadownictwa i Kwiaciarstwa, Oddział Pszczelnictwa
Machine Learning for Data Science (CS4786) Lecture11. Random Projections & Canonical Correlation Analysis
 Machine Learning for Data Science (CS4786) Lecture11 5 Random Projections & Canonical Correlation Analysis The Tall, THE FAT AND THE UGLY n X d The Tall, THE FAT AND THE UGLY d X > n X d n = n d d The
Machine Learning for Data Science (CS4786) Lecture11 5 Random Projections & Canonical Correlation Analysis The Tall, THE FAT AND THE UGLY n X d The Tall, THE FAT AND THE UGLY d X > n X d n = n d d The
Patients price acceptance SELECTED FINDINGS
 Patients price acceptance SELECTED FINDINGS October 2015 Summary With growing economy and Poles benefiting from this growth, perception of prices changes - this is also true for pharmaceuticals It may
Patients price acceptance SELECTED FINDINGS October 2015 Summary With growing economy and Poles benefiting from this growth, perception of prices changes - this is also true for pharmaceuticals It may
Rozpoznawanie twarzy metodą PCA Michał Bereta 1. Testowanie statystycznej istotności różnic między jakością klasyfikatorów
 Rozpoznawanie twarzy metodą PCA Michał Bereta www.michalbereta.pl 1. Testowanie statystycznej istotności różnic między jakością klasyfikatorów Wiemy, że możemy porównywad klasyfikatory np. za pomocą kroswalidacji.
Rozpoznawanie twarzy metodą PCA Michał Bereta www.michalbereta.pl 1. Testowanie statystycznej istotności różnic między jakością klasyfikatorów Wiemy, że możemy porównywad klasyfikatory np. za pomocą kroswalidacji.
 aforementioned device she also has to estimate the time when the patients need the infusion to be replaced and/or disconnected. Meanwhile, however, she must cope with many other tasks. If the department
aforementioned device she also has to estimate the time when the patients need the infusion to be replaced and/or disconnected. Meanwhile, however, she must cope with many other tasks. If the department
Machine Learning for Data Science (CS4786) Lecture 11. Spectral Embedding + Clustering
 Machine Learning for Data Science (CS4786) Lecture 11 Spectral Embedding + Clustering MOTIVATING EXAMPLE What can you say from this network? MOTIVATING EXAMPLE How about now? THOUGHT EXPERIMENT For each
Machine Learning for Data Science (CS4786) Lecture 11 Spectral Embedding + Clustering MOTIVATING EXAMPLE What can you say from this network? MOTIVATING EXAMPLE How about now? THOUGHT EXPERIMENT For each
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
MaPlan Sp. z O.O. Click here if your download doesn"t start automatically
 Mierzeja Wislana, mapa turystyczna 1:50 000: Mikoszewo, Jantar, Stegna, Sztutowo, Katy Rybackie, Przebrno, Krynica Morska, Piaski, Frombork =... = Carte touristique (Polish Edition) MaPlan Sp. z O.O Click
Mierzeja Wislana, mapa turystyczna 1:50 000: Mikoszewo, Jantar, Stegna, Sztutowo, Katy Rybackie, Przebrno, Krynica Morska, Piaski, Frombork =... = Carte touristique (Polish Edition) MaPlan Sp. z O.O Click
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
Has the heat wave frequency or intensity changed in Poland since 1950?
 Has the heat wave frequency or intensity changed in Poland since 1950? Joanna Wibig Department of Meteorology and Climatology, University of Lodz, Poland OUTLINE: Motivation Data Heat wave frequency measures
Has the heat wave frequency or intensity changed in Poland since 1950? Joanna Wibig Department of Meteorology and Climatology, University of Lodz, Poland OUTLINE: Motivation Data Heat wave frequency measures
ERASMUS + : Trail of extinct and active volcanoes, earthquakes through Europe. SURVEY TO STUDENTS.
 ERASMUS + : Trail of extinct and active volcanoes, earthquakes through Europe. SURVEY TO STUDENTS. Strona 1 1. Please give one answer. I am: Students involved in project 69% 18 Student not involved in
ERASMUS + : Trail of extinct and active volcanoes, earthquakes through Europe. SURVEY TO STUDENTS. Strona 1 1. Please give one answer. I am: Students involved in project 69% 18 Student not involved in
European Crime Prevention Award (ECPA) Annex I - new version 2014
 European Crime Prevention Award (ECPA) Annex I - new version 2014 Załącznik nr 1 General information (Informacje ogólne) 1. Please specify your country. (Kraj pochodzenia:) 2. Is this your country s ECPA
European Crime Prevention Award (ECPA) Annex I - new version 2014 Załącznik nr 1 General information (Informacje ogólne) 1. Please specify your country. (Kraj pochodzenia:) 2. Is this your country s ECPA
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
Zakopane, plan miasta: Skala ok. 1: = City map (Polish Edition)
 Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Click here if your download doesn"t start automatically Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Zakopane,
Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Click here if your download doesn"t start automatically Zakopane, plan miasta: Skala ok. 1:15 000 = City map (Polish Edition) Zakopane,
Revenue Maximization. Sept. 25, 2018
 Revenue Maximization Sept. 25, 2018 Goal So Far: Ideal Auctions Dominant-Strategy Incentive Compatible (DSIC) b i = v i is a dominant strategy u i 0 x is welfare-maximizing x and p run in polynomial time
Revenue Maximization Sept. 25, 2018 Goal So Far: Ideal Auctions Dominant-Strategy Incentive Compatible (DSIC) b i = v i is a dominant strategy u i 0 x is welfare-maximizing x and p run in polynomial time
Dominika Janik-Hornik (Uniwersytet Ekonomiczny w Katowicach) Kornelia Kamińska (ESN Akademia Górniczo-Hutnicza) Dorota Rytwińska (FRSE)
 Czy mobilność pracowników uczelni jest gwarancją poprawnej realizacji mobilności studentów? Jak polskie uczelnie wykorzystują mobilność pracowników w programie Erasmus+ do poprawiania stopnia umiędzynarodowienia
Czy mobilność pracowników uczelni jest gwarancją poprawnej realizacji mobilności studentów? Jak polskie uczelnie wykorzystują mobilność pracowników w programie Erasmus+ do poprawiania stopnia umiędzynarodowienia
HemoRec in Poland. Summary of bleeding episodes of haemophilia patients with inhibitor recorded in the years 2008 and 2009 04/2010
 HemoRec in Poland Summary of bleeding episodes of haemophilia patients with inhibitor recorded in the years 2008 and 2009 04/2010 Institute of Biostatistics and Analyses. Masaryk University. Brno Participating
HemoRec in Poland Summary of bleeding episodes of haemophilia patients with inhibitor recorded in the years 2008 and 2009 04/2010 Institute of Biostatistics and Analyses. Masaryk University. Brno Participating
Katowice, plan miasta: Skala 1: = City map = Stadtplan (Polish Edition)
 Katowice, plan miasta: Skala 1:20 000 = City map = Stadtplan (Polish Edition) Polskie Przedsiebiorstwo Wydawnictw Kartograficznych im. Eugeniusza Romera Click here if your download doesn"t start automatically
Katowice, plan miasta: Skala 1:20 000 = City map = Stadtplan (Polish Edition) Polskie Przedsiebiorstwo Wydawnictw Kartograficznych im. Eugeniusza Romera Click here if your download doesn"t start automatically
Unit of Social Gerontology, Institute of Labour and Social Studies ageing and its consequences for society
 Prof. Piotr Bledowski, Ph.D. Institute of Social Economy, Warsaw School of Economics local policy, social security, labour market Unit of Social Gerontology, Institute of Labour and Social Studies ageing
Prof. Piotr Bledowski, Ph.D. Institute of Social Economy, Warsaw School of Economics local policy, social security, labour market Unit of Social Gerontology, Institute of Labour and Social Studies ageing
Charakterystyka kliniczna chorych na raka jelita grubego
 Lek. Łukasz Głogowski Charakterystyka kliniczna chorych na raka jelita grubego Rozprawa na stopień doktora nauk medycznych Opiekun naukowy: Dr hab. n. med. Ewa Nowakowska-Zajdel Zakład Profilaktyki Chorób
Lek. Łukasz Głogowski Charakterystyka kliniczna chorych na raka jelita grubego Rozprawa na stopień doktora nauk medycznych Opiekun naukowy: Dr hab. n. med. Ewa Nowakowska-Zajdel Zakład Profilaktyki Chorób
Stargard Szczecinski i okolice (Polish Edition)
 Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz Click here if your download doesn"t start automatically Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz
Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz Click here if your download doesn"t start automatically Stargard Szczecinski i okolice (Polish Edition) Janusz Leszek Jurkiewicz
Baptist Church Records
 Baptist Church Records The Baptist religion was a religious minority in Poland, making it more difficult to know when and where records of this religion might be available. In an article from Rodziny,
Baptist Church Records The Baptist religion was a religious minority in Poland, making it more difficult to know when and where records of this religion might be available. In an article from Rodziny,
Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition)
 Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition) J Krupski Click here if your download doesn"t start automatically Karpacz, plan miasta 1:10 000: Panorama
Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition) J Krupski Click here if your download doesn"t start automatically Karpacz, plan miasta 1:10 000: Panorama
Egzamin maturalny z języka angielskiego na poziomie dwujęzycznym Rozmowa wstępna (wyłącznie dla egzaminującego)
 112 Informator o egzaminie maturalnym z języka angielskiego od roku szkolnego 2014/2015 2.6.4. Część ustna. Przykładowe zestawy zadań Przykładowe pytania do rozmowy wstępnej Rozmowa wstępna (wyłącznie
112 Informator o egzaminie maturalnym z języka angielskiego od roku szkolnego 2014/2015 2.6.4. Część ustna. Przykładowe zestawy zadań Przykładowe pytania do rozmowy wstępnej Rozmowa wstępna (wyłącznie
deep learning for NLP (5 lectures)
 TTIC 31210: Advanced Natural Language Processing Kevin Gimpel Spring 2019 Lecture 6: Finish Transformers; Sequence- to- Sequence Modeling and AJenKon 1 Roadmap intro (1 lecture) deep learning for NLP (5
TTIC 31210: Advanced Natural Language Processing Kevin Gimpel Spring 2019 Lecture 6: Finish Transformers; Sequence- to- Sequence Modeling and AJenKon 1 Roadmap intro (1 lecture) deep learning for NLP (5
EFFECT OF QUEEN CAGING CONDITIONS ON INSEMINATION RESULTS
 Vol. 49 No. 1 2005 Journal of Apicultural Science 5 EFFECT OF QUEEN CAGING CONDITIONS ON INSEMINATION RESULTS Aldona Gontarz 1, Ma³gorzata Bieñkowska 2, Krzysztof Loc 3 1 Department of breeding methods
Vol. 49 No. 1 2005 Journal of Apicultural Science 5 EFFECT OF QUEEN CAGING CONDITIONS ON INSEMINATION RESULTS Aldona Gontarz 1, Ma³gorzata Bieñkowska 2, Krzysztof Loc 3 1 Department of breeding methods
Proposal of thesis topic for mgr in. (MSE) programme in Telecommunications and Computer Science
 Proposal of thesis topic for mgr in (MSE) programme 1 Topic: Monte Carlo Method used for a prognosis of a selected technological process 2 Supervisor: Dr in Małgorzata Langer 3 Auxiliary supervisor: 4
Proposal of thesis topic for mgr in (MSE) programme 1 Topic: Monte Carlo Method used for a prognosis of a selected technological process 2 Supervisor: Dr in Małgorzata Langer 3 Auxiliary supervisor: 4
Miedzy legenda a historia: Szlakiem piastowskim z Poznania do Gniezna (Biblioteka Kroniki Wielkopolski) (Polish Edition)
 Miedzy legenda a historia: Szlakiem piastowskim z Poznania do Gniezna (Biblioteka Kroniki Wielkopolski) (Polish Edition) Piotr Maluskiewicz Click here if your download doesn"t start automatically Miedzy
Miedzy legenda a historia: Szlakiem piastowskim z Poznania do Gniezna (Biblioteka Kroniki Wielkopolski) (Polish Edition) Piotr Maluskiewicz Click here if your download doesn"t start automatically Miedzy
ABOUT NEW EASTERN EUROPE BESTmQUARTERLYmJOURNAL
 ABOUT NEW EASTERN EUROPE BESTmQUARTERLYmJOURNAL Formanminsidemlookmatmpoliticsxmculturexmsocietymandm economyminmthemregionmofmcentralmandmeasternm EuropexmtheremismnomothermsourcemlikemNew Eastern EuropeImSincemitsmlaunchminmPw--xmthemmagazinemhasm
ABOUT NEW EASTERN EUROPE BESTmQUARTERLYmJOURNAL Formanminsidemlookmatmpoliticsxmculturexmsocietymandm economyminmthemregionmofmcentralmandmeasternm EuropexmtheremismnomothermsourcemlikemNew Eastern EuropeImSincemitsmlaunchminmPw--xmthemmagazinemhasm
Pielgrzymka do Ojczyzny: Przemowienia i homilie Ojca Swietego Jana Pawla II (Jan Pawel II-- pierwszy Polak na Stolicy Piotrowej) (Polish Edition)
 Pielgrzymka do Ojczyzny: Przemowienia i homilie Ojca Swietego Jana Pawla II (Jan Pawel II-- pierwszy Polak na Stolicy Piotrowej) (Polish Edition) Click here if your download doesn"t start automatically
Pielgrzymka do Ojczyzny: Przemowienia i homilie Ojca Swietego Jana Pawla II (Jan Pawel II-- pierwszy Polak na Stolicy Piotrowej) (Polish Edition) Click here if your download doesn"t start automatically
Hard-Margin Support Vector Machines
 Hard-Margin Support Vector Machines aaacaxicbzdlssnafiyn9vbjlepk3ay2gicupasvu4iblxuaw2hjmuwn7ddjjmxm1bkcg1/fjqsvt76fo9/gazqfvn8y+pjpozw5vx8zkpvtfxmlhcwl5zxyqrm2vrg5zw3vxmsoezi4ogkr6phieky5crvvjhriqvdom9l2xxftevuwcekj3lktmhghgniauiyutvrwxtvme34a77kbvg73gtygpjsrfati1+xc8c84bvraowbf+uwnipyehcvmkjrdx46vlykhkgykm3ujjdhcyzqkxy0chur6ax5cbg+1m4bbjptjcubuz4kuhvjoql93hkin5hxtav5x6yyqopnsyuneey5ni4keqrxbar5wqaxbik00icyo/iveiyqqvjo1u4fgzj/8f9x67bzmxnurjzmijtlybwfgcdjgfdtajwgcf2dwaj7ac3g1ho1n4814n7wwjgjmf/ys8fenfycuzq==
Hard-Margin Support Vector Machines aaacaxicbzdlssnafiyn9vbjlepk3ay2gicupasvu4iblxuaw2hjmuwn7ddjjmxm1bkcg1/fjqsvt76fo9/gazqfvn8y+pjpozw5vx8zkpvtfxmlhcwl5zxyqrm2vrg5zw3vxmsoezi4ogkr6phieky5crvvjhriqvdom9l2xxftevuwcekj3lktmhghgniauiyutvrwxtvme34a77kbvg73gtygpjsrfati1+xc8c84bvraowbf+uwnipyehcvmkjrdx46vlykhkgykm3ujjdhcyzqkxy0chur6ax5cbg+1m4bbjptjcubuz4kuhvjoql93hkin5hxtav5x6yyqopnsyuneey5ni4keqrxbar5wqaxbik00icyo/iveiyqqvjo1u4fgzj/8f9x67bzmxnurjzmijtlybwfgcdjgfdtajwgcf2dwaj7ac3g1ho1n4814n7wwjgjmf/ys8fenfycuzq==
Uniwersytet Medyczny w Łodzi. Wydział Lekarski. Jarosław Woźniak. Rozprawa doktorska
 Uniwersytet Medyczny w Łodzi Wydział Lekarski Jarosław Woźniak Rozprawa doktorska Ocena funkcji stawu skokowego po leczeniu operacyjnym złamań kostek goleni z uszkodzeniem więzozrostu piszczelowo-strzałkowego
Uniwersytet Medyczny w Łodzi Wydział Lekarski Jarosław Woźniak Rozprawa doktorska Ocena funkcji stawu skokowego po leczeniu operacyjnym złamań kostek goleni z uszkodzeniem więzozrostu piszczelowo-strzałkowego
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Raport bieżący: 44/2018 Data: g. 21:03 Skrócona nazwa emitenta: SERINUS ENERGY plc
 Raport bieżący: 44/2018 Data: 2018-05-23 g. 21:03 Skrócona nazwa emitenta: SERINUS ENERGY plc Temat: Zawiadomienie o zmianie udziału w ogólnej liczbie głosów w Serinus Energy plc Podstawa prawna: Inne
Raport bieżący: 44/2018 Data: 2018-05-23 g. 21:03 Skrócona nazwa emitenta: SERINUS ENERGY plc Temat: Zawiadomienie o zmianie udziału w ogólnej liczbie głosów w Serinus Energy plc Podstawa prawna: Inne
Łukasz Reszka Wiceprezes Zarządu
 Łukasz Reszka Wiceprezes Zarządu Time for changes! Vocational activisation young unemployed people aged 15 to 24 Projekt location Ząbkowice Śląskie project produced in cooperation with Poviat Labour Office
Łukasz Reszka Wiceprezes Zarządu Time for changes! Vocational activisation young unemployed people aged 15 to 24 Projekt location Ząbkowice Śląskie project produced in cooperation with Poviat Labour Office
Wstęp ARTYKUŁ REDAKCYJNY / LEADING ARTICLE
 Dzieciństwo w cieniu schizofrenii przegląd literatury na temat możliwych form pomocy i wsparcia dzieci z rodzin, gdzie jeden z rodziców dotknięty jest schizofrenią Childhood in the shadow of schizophrenia
Dzieciństwo w cieniu schizofrenii przegląd literatury na temat możliwych form pomocy i wsparcia dzieci z rodzin, gdzie jeden z rodziców dotknięty jest schizofrenią Childhood in the shadow of schizophrenia
Extraclass. Football Men. Season 2009/10 - Autumn round
 Extraclass Football Men Season 2009/10 - Autumn round Invitation Dear All, On the date of 29th July starts the new season of Polish Extraclass. There will be live coverage form all the matches on Canal+
Extraclass Football Men Season 2009/10 - Autumn round Invitation Dear All, On the date of 29th July starts the new season of Polish Extraclass. There will be live coverage form all the matches on Canal+
ROZPRAWA DOKTORSKA. Mateusz Romanowski
 Mateusz Romanowski Wpływ krioterapii ogólnoustrojowej na aktywność choroby i sprawność chorych na zesztywniające zapalenie stawów kręgosłupa ROZPRAWA DOKTORSKA Promotor: Dr hab., prof. AWF Anna Straburzyńska-Lupa
Mateusz Romanowski Wpływ krioterapii ogólnoustrojowej na aktywność choroby i sprawność chorych na zesztywniające zapalenie stawów kręgosłupa ROZPRAWA DOKTORSKA Promotor: Dr hab., prof. AWF Anna Straburzyńska-Lupa
BIOPHYSICS. Politechnika Łódzka, ul. Żeromskiego 116, Łódź, tel. (042)
 BIOPHYSICS Prezentacja multimedialna współfinansowana przez Unię Europejską w ramach Europejskiego Funduszu Społecznego w projekcie pt. Innowacyjna dydaktyka bez ograniczeń - zintegrowany rozwój Politechniki
BIOPHYSICS Prezentacja multimedialna współfinansowana przez Unię Europejską w ramach Europejskiego Funduszu Społecznego w projekcie pt. Innowacyjna dydaktyka bez ograniczeń - zintegrowany rozwój Politechniki
Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition)
 Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition) J Krupski Click here if your download doesn"t start automatically Karpacz, plan miasta 1:10 000: Panorama
Karpacz, plan miasta 1:10 000: Panorama Karkonoszy, mapa szlakow turystycznych (Polish Edition) J Krupski Click here if your download doesn"t start automatically Karpacz, plan miasta 1:10 000: Panorama
Krytyczne czynniki sukcesu w zarządzaniu projektami
 Seweryn SPAŁEK Krytyczne czynniki sukcesu w zarządzaniu projektami MONOGRAFIA Wydawnictwo Politechniki Śląskiej Gliwice 2004 SPIS TREŚCI WPROWADZENIE 5 1. ZARZĄDZANIE PROJEKTAMI W ORGANIZACJI 13 1.1. Zarządzanie
Seweryn SPAŁEK Krytyczne czynniki sukcesu w zarządzaniu projektami MONOGRAFIA Wydawnictwo Politechniki Śląskiej Gliwice 2004 SPIS TREŚCI WPROWADZENIE 5 1. ZARZĄDZANIE PROJEKTAMI W ORGANIZACJI 13 1.1. Zarządzanie
Konsorcjum Śląskich Uczelni Publicznych
 Konsorcjum Śląskich Uczelni Publicznych Dlaczego powstało? - świat przeżywa dziś rewolucję w obszarze edukacji, - naszym celem jest promocja śląskiego jako regionu opartego na wiedzy, i najnowszych technologiach,
Konsorcjum Śląskich Uczelni Publicznych Dlaczego powstało? - świat przeżywa dziś rewolucję w obszarze edukacji, - naszym celem jest promocja śląskiego jako regionu opartego na wiedzy, i najnowszych technologiach,
Dr hab. Beata Madras-Majewska, prof. SGGW
 Dr hab. Beata Madras-Majewska, prof. SGGW KONTAKT SGGW w Warszawie Wydział Nauk o Zwierzętach Pracownia Pszczelnictwa ul. Nowoursynowska 166 02-787 Warszawa Tel: + 48 225936652 E-mail: beata_madras_majewska@sggw.pl
Dr hab. Beata Madras-Majewska, prof. SGGW KONTAKT SGGW w Warszawie Wydział Nauk o Zwierzętach Pracownia Pszczelnictwa ul. Nowoursynowska 166 02-787 Warszawa Tel: + 48 225936652 E-mail: beata_madras_majewska@sggw.pl
EXAMPLES OF CABRI GEOMETRE II APPLICATION IN GEOMETRIC SCIENTIFIC RESEARCH
 Anna BŁACH Centre of Geometry and Engineering Graphics Silesian University of Technology in Gliwice EXAMPLES OF CABRI GEOMETRE II APPLICATION IN GEOMETRIC SCIENTIFIC RESEARCH Introduction Computer techniques
Anna BŁACH Centre of Geometry and Engineering Graphics Silesian University of Technology in Gliwice EXAMPLES OF CABRI GEOMETRE II APPLICATION IN GEOMETRIC SCIENTIFIC RESEARCH Introduction Computer techniques
Jak zasada Pareto może pomóc Ci w nauce języków obcych?
 Jak zasada Pareto może pomóc Ci w nauce języków obcych? Artykuł pobrano ze strony eioba.pl Pokazuje, jak zastosowanie zasady Pareto może usprawnić Twoją naukę angielskiego. Słynna zasada Pareto mówi o
Jak zasada Pareto może pomóc Ci w nauce języków obcych? Artykuł pobrano ze strony eioba.pl Pokazuje, jak zastosowanie zasady Pareto może usprawnić Twoją naukę angielskiego. Słynna zasada Pareto mówi o
Podstawa prawna: Art. 70 pkt 1 Ustawy o ofercie - nabycie lub zbycie znacznego pakietu akcji
 Raport bieżący: 41/2018 Data: 2018-05-22 g. 08:01 Skrócona nazwa emitenta: SERINUS ENERGY plc Temat: Przekroczenie progu 5% głosów w SERINUS ENERGY plc Podstawa prawna: Art. 70 pkt 1 Ustawy o ofercie -
Raport bieżący: 41/2018 Data: 2018-05-22 g. 08:01 Skrócona nazwa emitenta: SERINUS ENERGY plc Temat: Przekroczenie progu 5% głosów w SERINUS ENERGY plc Podstawa prawna: Art. 70 pkt 1 Ustawy o ofercie -
Latent Dirichlet Allocation Models and their Evaluation IT for Practice 2016
 Latent Dirichlet Allocation Models and their Evaluation IT for Practice 2016 Paweł Lula Cracow University of Economics, Poland pawel.lula@uek.krakow.pl Latent Dirichlet Allocation (LDA) Documents Latent
Latent Dirichlet Allocation Models and their Evaluation IT for Practice 2016 Paweł Lula Cracow University of Economics, Poland pawel.lula@uek.krakow.pl Latent Dirichlet Allocation (LDA) Documents Latent
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Cracow University of Economics Poland. Overview. Sources of Real GDP per Capita Growth: Polish Regional-Macroeconomic Dimensions 2000-2005
 Cracow University of Economics Sources of Real GDP per Capita Growth: Polish Regional-Macroeconomic Dimensions 2000-2005 - Key Note Speech - Presented by: Dr. David Clowes The Growth Research Unit CE Europe
Cracow University of Economics Sources of Real GDP per Capita Growth: Polish Regional-Macroeconomic Dimensions 2000-2005 - Key Note Speech - Presented by: Dr. David Clowes The Growth Research Unit CE Europe
ZGŁOSZENIE WSPÓLNEGO POLSKO -. PROJEKTU NA LATA: APPLICATION FOR A JOINT POLISH -... PROJECT FOR THE YEARS:.
 ZGŁOSZENIE WSPÓLNEGO POLSKO -. PROJEKTU NA LATA: APPLICATION FOR A JOINT POLISH -... PROJECT FOR THE YEARS:. W RAMACH POROZUMIENIA O WSPÓŁPRACY NAUKOWEJ MIĘDZY POLSKĄ AKADEMIĄ NAUK I... UNDER THE AGREEMENT
ZGŁOSZENIE WSPÓLNEGO POLSKO -. PROJEKTU NA LATA: APPLICATION FOR A JOINT POLISH -... PROJECT FOR THE YEARS:. W RAMACH POROZUMIENIA O WSPÓŁPRACY NAUKOWEJ MIĘDZY POLSKĄ AKADEMIĄ NAUK I... UNDER THE AGREEMENT
Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2)
 Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Click here if your download doesn"t start automatically Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Emilka szuka swojej gwiazdy / Emily
Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Click here if your download doesn"t start automatically Emilka szuka swojej gwiazdy / Emily Climbs (Emily, #2) Emilka szuka swojej gwiazdy / Emily
DM-ML, DM-FL. Auxiliary Equipment and Accessories. Damper Drives. Dimensions. Descritpion
 DM-ML, DM-FL Descritpion DM-ML and DM-FL actuators are designed for driving round dampers and square multi-blade dampers. Example identification Product code: DM-FL-5-2 voltage Dimensions DM-ML-6 DM-ML-8
DM-ML, DM-FL Descritpion DM-ML and DM-FL actuators are designed for driving round dampers and square multi-blade dampers. Example identification Product code: DM-FL-5-2 voltage Dimensions DM-ML-6 DM-ML-8
Domy inaczej pomyślane A different type of housing CEZARY SANKOWSKI
 Domy inaczej pomyślane A different type of housing CEZARY SANKOWSKI O tym, dlaczego warto budować pasywnie, komu budownictwo pasywne się opłaca, a kto się go boi, z architektem, Cezarym Sankowskim, rozmawia
Domy inaczej pomyślane A different type of housing CEZARY SANKOWSKI O tym, dlaczego warto budować pasywnie, komu budownictwo pasywne się opłaca, a kto się go boi, z architektem, Cezarym Sankowskim, rozmawia
Wydział Fizyki, Astronomii i Informatyki Stosowanej Uniwersytet Mikołaja Kopernika w Toruniu
 IONS-14 / OPTO Meeting For Young Researchers 2013 Khet Tournament On 3-6 July 2013 at the Faculty of Physics, Astronomy and Informatics of Nicolaus Copernicus University in Torun (Poland) there were two
IONS-14 / OPTO Meeting For Young Researchers 2013 Khet Tournament On 3-6 July 2013 at the Faculty of Physics, Astronomy and Informatics of Nicolaus Copernicus University in Torun (Poland) there were two
Zarządzanie sieciami telekomunikacyjnymi
 SNMP Protocol The Simple Network Management Protocol (SNMP) is an application layer protocol that facilitates the exchange of management information between network devices. It is part of the Transmission
SNMP Protocol The Simple Network Management Protocol (SNMP) is an application layer protocol that facilitates the exchange of management information between network devices. It is part of the Transmission
Ocena potrzeb pacjentów z zaburzeniami psychicznymi
 Mikołaj Trizna Ocena potrzeb pacjentów z zaburzeniami psychicznymi przebywających na oddziałach psychiatrii sądowej Rozprawa na stopień doktora nauk medycznych Promotor: dr hab.n.med. Tomasz Adamowski,
Mikołaj Trizna Ocena potrzeb pacjentów z zaburzeniami psychicznymi przebywających na oddziałach psychiatrii sądowej Rozprawa na stopień doktora nauk medycznych Promotor: dr hab.n.med. Tomasz Adamowski,
Sargent Opens Sonairte Farmers' Market
 Sargent Opens Sonairte Farmers' Market 31 March, 2008 1V8VIZSV7EVKIRX8(1MRMWXIVSJ7XEXIEXXLI(ITEVXQIRXSJ%KVMGYPXYVI *MWLIVMIWERH*SSHTIVJSVQIHXLISJJMGMEPSTIRMRKSJXLI7SREMVXI*EVQIVW 1EVOIXMR0E]XS[R'S1IEXL
Sargent Opens Sonairte Farmers' Market 31 March, 2008 1V8VIZSV7EVKIRX8(1MRMWXIVSJ7XEXIEXXLI(ITEVXQIRXSJ%KVMGYPXYVI *MWLIVMIWERH*SSHTIVJSVQIHXLISJJMGMEPSTIRMRKSJXLI7SREMVXI*EVQIVW 1EVOIXMR0E]XS[R'S1IEXL
BSSSC Baltic Sea States Subregional Co-operation. operation good practices presentation
 BSSSC Baltic Sea States Subregional Co-operation operation good practices presentation Polites association was founded in 2002 in Szczecin Stowarzyszenie POLITES w Szczecinie Starszy Brat Starsza Siostra
BSSSC Baltic Sea States Subregional Co-operation operation good practices presentation Polites association was founded in 2002 in Szczecin Stowarzyszenie POLITES w Szczecinie Starszy Brat Starsza Siostra
Umowa Licencyjna Użytkownika Końcowego End-user licence agreement
 Umowa Licencyjna Użytkownika Końcowego End-user licence agreement Umowa Licencyjna Użytkownika Końcowego Wersja z dnia 2 września 2014 Definicje GRA - Przeglądarkowa gra HTML5 o nazwie Sumerian City, dostępna
Umowa Licencyjna Użytkownika Końcowego End-user licence agreement Umowa Licencyjna Użytkownika Końcowego Wersja z dnia 2 września 2014 Definicje GRA - Przeglądarkowa gra HTML5 o nazwie Sumerian City, dostępna
Ankiety Nowe funkcje! Pomoc magda.szewczyk@slo-wroc.pl. magda.szewczyk@slo-wroc.pl. Twoje konto Wyloguj. BIODIVERSITY OF RIVERS: Survey to students
 Ankiety Nowe funkcje! Pomoc magda.szewczyk@slo-wroc.pl Back Twoje konto Wyloguj magda.szewczyk@slo-wroc.pl BIODIVERSITY OF RIVERS: Survey to students Tworzenie ankiety Udostępnianie Analiza (55) Wyniki
Ankiety Nowe funkcje! Pomoc magda.szewczyk@slo-wroc.pl Back Twoje konto Wyloguj magda.szewczyk@slo-wroc.pl BIODIVERSITY OF RIVERS: Survey to students Tworzenie ankiety Udostępnianie Analiza (55) Wyniki
 !850016! www.irs.gov/form8879eo. e-file www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C,
!850016! www.irs.gov/form8879eo. e-file www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C,
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
SWPS Uniwersytet Humanistycznospołeczny. Wydział Zamiejscowy we Wrocławiu. Karolina Horodyska
 SWPS Uniwersytet Humanistycznospołeczny Wydział Zamiejscowy we Wrocławiu Karolina Horodyska Warunki skutecznego promowania zdrowej diety i aktywności fizycznej: dobre praktyki w interwencjach psychospołecznych
SWPS Uniwersytet Humanistycznospołeczny Wydział Zamiejscowy we Wrocławiu Karolina Horodyska Warunki skutecznego promowania zdrowej diety i aktywności fizycznej: dobre praktyki w interwencjach psychospołecznych
Installation of EuroCert software for qualified electronic signature
 Installation of EuroCert software for qualified electronic signature for Microsoft Windows systems Warsaw 28.08.2019 Content 1. Downloading and running the software for the e-signature... 3 a) Installer
Installation of EuroCert software for qualified electronic signature for Microsoft Windows systems Warsaw 28.08.2019 Content 1. Downloading and running the software for the e-signature... 3 a) Installer
photo graphic Jan Witkowski Project for exhibition compositions typography colors : +48 506 780 943 : janwi@janwi.com
 Jan Witkowski : +48 506 780 943 : janwi@janwi.com Project for exhibition photo graphic compositions typography colors Berlin London Paris Barcelona Vienna Prague Krakow Zakopane Jan Witkowski ARTIST FROM
Jan Witkowski : +48 506 780 943 : janwi@janwi.com Project for exhibition photo graphic compositions typography colors Berlin London Paris Barcelona Vienna Prague Krakow Zakopane Jan Witkowski ARTIST FROM
Network Services for Spatial Data in European Geo-Portals and their Compliance with ISO and OGC Standards
 INSPIRE Conference 2010 INSPIRE as a Framework for Cooperation Network Services for Spatial Data in European Geo-Portals and their Compliance with ISO and OGC Standards Elżbieta Bielecka Agnieszka Zwirowicz
INSPIRE Conference 2010 INSPIRE as a Framework for Cooperation Network Services for Spatial Data in European Geo-Portals and their Compliance with ISO and OGC Standards Elżbieta Bielecka Agnieszka Zwirowicz
SubVersion. Piotr Mikulski. SubVersion. P. Mikulski. Co to jest subversion? Zalety SubVersion. Wady SubVersion. Inne różnice SubVersion i CVS
 Piotr Mikulski 2006 Subversion is a free/open-source version control system. That is, Subversion manages files and directories over time. A tree of files is placed into a central repository. The repository
Piotr Mikulski 2006 Subversion is a free/open-source version control system. That is, Subversion manages files and directories over time. A tree of files is placed into a central repository. The repository
DOI: / /32/37
 . 2015. 4 (32) 1:18 DOI: 10.17223/1998863 /32/37 -,,. - -. :,,,,., -, -.,.-.,.,.,. -., -,.,,., -, 70 80. (.,.,. ),, -,.,, -,, (1886 1980).,.,, (.,.,..), -, -,,,, ; -, - 346, -,.. :, -, -,,,,,.,,, -,,,
. 2015. 4 (32) 1:18 DOI: 10.17223/1998863 /32/37 -,,. - -. :,,,,., -, -.,.-.,.,.,. -., -,.,,., -, 70 80. (.,.,. ),, -,.,, -,, (1886 1980).,.,, (.,.,..), -, -,,,, ; -, - 346, -,.. :, -, -,,,,,.,,, -,,,
Pro-tumoral immune cell alterations in wild type and Shbdeficient mice in response to 4T1 breast carcinomas
 www.oncotarget.com Oncotarget, Supplementary Materials Pro-tumoral immune cell alterations in wild type and Shbdeficient mice in response to 4T1 breast carcinomas SUPPLEMENTARY MATERIALS Supplementary
www.oncotarget.com Oncotarget, Supplementary Materials Pro-tumoral immune cell alterations in wild type and Shbdeficient mice in response to 4T1 breast carcinomas SUPPLEMENTARY MATERIALS Supplementary
Zajęcia z języka angielskiego TELC Gimnazjum Scenariusz lekcji Prowadzący: Jarosław Gołębiewski Temat: Czas Present Perfect - wprowadzenie
 Zajęcia z języka angielskiego TELC Gimnazjum Scenariusz lekcji Prowadzący: Jarosław Gołębiewski Temat: Czas Present Perfect - wprowadzenie I. Cele lekcji 1) Wiadomości Uczeń: wie, że czas present perfect
Zajęcia z języka angielskiego TELC Gimnazjum Scenariusz lekcji Prowadzący: Jarosław Gołębiewski Temat: Czas Present Perfect - wprowadzenie I. Cele lekcji 1) Wiadomości Uczeń: wie, że czas present perfect
Gdański Uniwersytet Medyczny Wydział Nauk o Zdrowiu z Oddziałem Pielęgniarstwa i Instytutem Medycyny Morskiej i Tropikalnej. Beata Wieczorek-Wójcik
 Gdański Uniwersytet Medyczny Wydział Nauk o Zdrowiu z Oddziałem Pielęgniarstwa i Instytutem Medycyny Morskiej i Tropikalnej Beata Wieczorek-Wójcik Poziom obsad pielęgniarskich a częstość i rodzaj zdarzeń
Gdański Uniwersytet Medyczny Wydział Nauk o Zdrowiu z Oddziałem Pielęgniarstwa i Instytutem Medycyny Morskiej i Tropikalnej Beata Wieczorek-Wójcik Poziom obsad pielęgniarskich a częstość i rodzaj zdarzeń
Evaluation of the main goal and specific objectives of the Human Capital Operational Programme
 Pracownia Naukowo-Edukacyjna Evaluation of the main goal and specific objectives of the Human Capital Operational Programme and the contribution by ESF funds towards the results achieved within specific
Pracownia Naukowo-Edukacyjna Evaluation of the main goal and specific objectives of the Human Capital Operational Programme and the contribution by ESF funds towards the results achieved within specific
TRANSPORT W RODZINNYCH GOSPODARSTWACH ROLNYCH
 INŻYNIERIA W ROLNICTWIE. MONOGRAFIE 16 ENGINEERING IN AGRICULTURE. MONOGRAPHS 16 WIESŁAW GOLKA TRANSPORT W RODZINNYCH GOSPODARSTWACH ROLNYCH TRANSPORTATION IN RURAL FAMILY FARMS Falenty 2014 WYDAWNICTWO
INŻYNIERIA W ROLNICTWIE. MONOGRAFIE 16 ENGINEERING IN AGRICULTURE. MONOGRAPHS 16 WIESŁAW GOLKA TRANSPORT W RODZINNYCH GOSPODARSTWACH ROLNYCH TRANSPORTATION IN RURAL FAMILY FARMS Falenty 2014 WYDAWNICTWO
OpenPoland.net API Documentation
 OpenPoland.net API Documentation Release 1.0 Michał Gryczka July 11, 2014 Contents 1 REST API tokens: 3 1.1 How to get a token............................................ 3 2 REST API : search for assets
OpenPoland.net API Documentation Release 1.0 Michał Gryczka July 11, 2014 Contents 1 REST API tokens: 3 1.1 How to get a token............................................ 3 2 REST API : search for assets
Few-fermion thermometry
 Few-fermion thermometry Phys. Rev. A 97, 063619 (2018) Tomasz Sowiński Institute of Physics of the Polish Academy of Sciences Co-authors: Marcin Płodzień Rafał Demkowicz-Dobrzański FEW-BODY PROBLEMS FewBody.ifpan.edu.pl
Few-fermion thermometry Phys. Rev. A 97, 063619 (2018) Tomasz Sowiński Institute of Physics of the Polish Academy of Sciences Co-authors: Marcin Płodzień Rafał Demkowicz-Dobrzański FEW-BODY PROBLEMS FewBody.ifpan.edu.pl
Agata i Rados aw Kowalscy
 Agata i Rados aw Kowalscy Akwakultura s odkowodna trend zmian na przestrzeni 21 wieku Akwakultura s odkowodna ca kowita produkcja w 2013r w przeliczeniu na area dost pny 1000000 900000 Produkcja ryb w
Agata i Rados aw Kowalscy Akwakultura s odkowodna trend zmian na przestrzeni 21 wieku Akwakultura s odkowodna ca kowita produkcja w 2013r w przeliczeniu na area dost pny 1000000 900000 Produkcja ryb w
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition)
 Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
Wojewodztwo Koszalinskie: Obiekty i walory krajoznawcze (Inwentaryzacja krajoznawcza Polski) (Polish Edition) Robert Respondowski Click here if your download doesn"t start automatically Wojewodztwo Koszalinskie:
PSZCZELNICZE ZESZYTY NAUKOWE Rok XXXVII 1993
 PSZCZELNICZE ZESZYTY NAUKOWE Rok XXXVII 1993 PODEJMOWANIE CZERWIENIA PRZEZ MATKI PSZCZELE PRZETRZYMYWANE W SKRZYNKACH W ASYŚCIE SWOBODNIE OBLATUJĄCYCH SIĘ PSZCZÓŁ B o ż e n a C h u d a - M i c k i e w
PSZCZELNICZE ZESZYTY NAUKOWE Rok XXXVII 1993 PODEJMOWANIE CZERWIENIA PRZEZ MATKI PSZCZELE PRZETRZYMYWANE W SKRZYNKACH W ASYŚCIE SWOBODNIE OBLATUJĄCYCH SIĘ PSZCZÓŁ B o ż e n a C h u d a - M i c k i e w
 www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
www.irs.gov/form990. If "Yes," complete Schedule A Schedule B, Schedule of Contributors If "Yes," complete Schedule C, Part I If "Yes," complete Schedule C, Part II If "Yes," complete Schedule C, Part
XT001_ INTRODUCTION TO EXIT INTERVIEW PYTANIE NIE JEST ZADAWANE W POLSCE W 2006 ROKU. WCIŚNIJ Ctrl+R BY PRZEJŚĆ DALEJ. 1.
 Share w2 Exit Questionnaire version 2.7 2006-09-29 XT001_ INTRODUCTION TO EXIT INTERVIEW 1. Kontynuuj XT006_ PROXY RESPONDENT'S SEX 1. Mężczyzna 2. Kobieta XT002_ RELATIONSHIP TO THE DECEASED IF XT002_
Share w2 Exit Questionnaire version 2.7 2006-09-29 XT001_ INTRODUCTION TO EXIT INTERVIEW 1. Kontynuuj XT006_ PROXY RESPONDENT'S SEX 1. Mężczyzna 2. Kobieta XT002_ RELATIONSHIP TO THE DECEASED IF XT002_
Akademia Morska w Szczecinie. Wydział Mechaniczny
 Akademia Morska w Szczecinie Wydział Mechaniczny ROZPRAWA DOKTORSKA mgr inż. Marcin Kołodziejski Analiza metody obsługiwania zarządzanego niezawodnością pędników azymutalnych platformy pływającej Promotor:
Akademia Morska w Szczecinie Wydział Mechaniczny ROZPRAWA DOKTORSKA mgr inż. Marcin Kołodziejski Analiza metody obsługiwania zarządzanego niezawodnością pędników azymutalnych platformy pływającej Promotor:
Nazwa projektu: Kreatywni i innowacyjni uczniowie konkurencyjni na rynku pracy
 Nazwa projektu: Kreatywni i innowacyjni uczniowie konkurencyjni na rynku pracy DZIAŁANIE 3.2 EDUKACJA OGÓLNA PODDZIAŁANIE 3.2.1 JAKOŚĆ EDUKACJI OGÓLNEJ Projekt współfinansowany przez Unię Europejską w
Nazwa projektu: Kreatywni i innowacyjni uczniowie konkurencyjni na rynku pracy DZIAŁANIE 3.2 EDUKACJA OGÓLNA PODDZIAŁANIE 3.2.1 JAKOŚĆ EDUKACJI OGÓLNEJ Projekt współfinansowany przez Unię Europejską w
Towards Stability Analysis of Data Transport Mechanisms: a Fluid Model and an Application
 Towards Stability Analysis of Data Transport Mechanisms: a Fluid Model and an Application Gayane Vardoyan *, C. V. Hollot, Don Towsley* * College of Information and Computer Sciences, Department of Electrical
Towards Stability Analysis of Data Transport Mechanisms: a Fluid Model and an Application Gayane Vardoyan *, C. V. Hollot, Don Towsley* * College of Information and Computer Sciences, Department of Electrical
Camspot 4.4 Camspot 4.5
 User manual (addition) Dodatek do instrukcji obsługi Camspot 4.4 Camspot 4.5 1. WiFi configuration 2. Configuration of sending pictures to e-mail/ftp after motion detection 1. Konfiguracja WiFi 2. Konfiguracja
User manual (addition) Dodatek do instrukcji obsługi Camspot 4.4 Camspot 4.5 1. WiFi configuration 2. Configuration of sending pictures to e-mail/ftp after motion detection 1. Konfiguracja WiFi 2. Konfiguracja
Is there a relationship between age and side dominance of tubal ectopic pregnancies? A preliminary report
 Is there a relationship between age and side dominance of tubal ectopic pregnancies? A preliminary report Czy istnieje zależność pomiędzy wiekiem i stroną, po której umiejscawia się ciąża ektopowa jajowodowa?
Is there a relationship between age and side dominance of tubal ectopic pregnancies? A preliminary report Czy istnieje zależność pomiędzy wiekiem i stroną, po której umiejscawia się ciąża ektopowa jajowodowa?
Employment. Number of employees employed on a contract of employment by gender in 2012. Company
 Im not found /sites/eneacsr2012.mess-asp.com/themes/eneacsr2012/img/enea.jpg Employt Capital Group is one of the largest companies in the energy industry. Therefore it has an influence, as an employer,
Im not found /sites/eneacsr2012.mess-asp.com/themes/eneacsr2012/img/enea.jpg Employt Capital Group is one of the largest companies in the energy industry. Therefore it has an influence, as an employer,
you see decision. oznacza to, Whenever kiedy widzisz biznes, someone once made Za każdym razem, który odnosi sukces,
 PREMIUM Za każdym razem, kiedy widzisz biznes, który odnosi sukces, oznacza to, że ktoś kiedyś podjął odważną decyzję. Whenever you see a successful business, someone once made a courageous decision. Szanowni
PREMIUM Za każdym razem, kiedy widzisz biznes, który odnosi sukces, oznacza to, że ktoś kiedyś podjął odważną decyzję. Whenever you see a successful business, someone once made a courageous decision. Szanowni
EPS. Erasmus Policy Statement
 Wyższa Szkoła Biznesu i Przedsiębiorczości Ostrowiec Świętokrzyski College of Business and Entrepreneurship EPS Erasmus Policy Statement Deklaracja Polityki Erasmusa 2014-2020 EN The institution is located
Wyższa Szkoła Biznesu i Przedsiębiorczości Ostrowiec Świętokrzyski College of Business and Entrepreneurship EPS Erasmus Policy Statement Deklaracja Polityki Erasmusa 2014-2020 EN The institution is located
Industrial Landscape Eustachy Kossakowski, 1950s and 60s
 dimensions: 38.5 x 28.5 cm technique: 12 black and white silver gelatin fiber prints made from original Eustachy Kossakowski s negatives edition: 6 price: 12 000 PLN Each portfolio is numbered and signed
dimensions: 38.5 x 28.5 cm technique: 12 black and white silver gelatin fiber prints made from original Eustachy Kossakowski s negatives edition: 6 price: 12 000 PLN Each portfolio is numbered and signed
Working Tax Credit Child Tax Credit Jobseeker s Allowance
 Benefits Depending on your residency status (EU citizen or not) there are various benefits available to help you with costs of living. A8 nationals need to have been working for a year and be registered
Benefits Depending on your residency status (EU citizen or not) there are various benefits available to help you with costs of living. A8 nationals need to have been working for a year and be registered
ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA JEZYKOWA) BY DOUGLAS KENT HALL
 Read Online and Download Ebook ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA JEZYKOWA) BY DOUGLAS KENT HALL DOWNLOAD EBOOK : ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA Click link bellow and free register
Read Online and Download Ebook ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA JEZYKOWA) BY DOUGLAS KENT HALL DOWNLOAD EBOOK : ARNOLD. EDUKACJA KULTURYSTY (POLSKA WERSJA Click link bellow and free register
Metodyki projektowania i modelowania systemów Cyganek & Kasperek & Rajda 2013 Katedra Elektroniki AGH
 Kierunek Elektronika i Telekomunikacja, Studia II stopnia Specjalność: Systemy wbudowane Metodyki projektowania i modelowania systemów Cyganek & Kasperek & Rajda 2013 Katedra Elektroniki AGH Zagadnienia
Kierunek Elektronika i Telekomunikacja, Studia II stopnia Specjalność: Systemy wbudowane Metodyki projektowania i modelowania systemów Cyganek & Kasperek & Rajda 2013 Katedra Elektroniki AGH Zagadnienia
